request an appointment online.
- Diagnosis & Treatment
- Cancer Types
- Non-Hodgkin Lymphoma
- Non-Hodgkin Lymphoma Treatment
Get details about our clinical trials that are currently enrolling patients.
View Clinical TrialsTreatment for Non-Hodgkin Lymphoma
UT MD Anderson is committed to helping people with non-Hodgkin lymphoma live longer, healthier lives — and we’re making great strides toward advanced therapies that minimize side effects.
Our Lymphoma and Myeloma Center has shaped the way lymphoma and myeloma are treated throughout the world.
We were leaders in many of the clinical trials that led to the treatments that are commonly used in lymphoma. The clinical trials that our group lead now may provide early access to treatments that eventually become the best new treatments for tomorrow.
We have found the most successful way to treat indolent (slow-growing) Non-Hodgkin lymphoma often is with strategies that limit or avoid the use of chemotherapy and have less impact on your body.
If you are diagnosed with non-Hodgkin lymphoma, your doctor will discuss the best options to treat it. This depends on several factors, including:
- Type of lymphoma
- Stage and category of disease
- Symptoms
- Your age and general health
Every lymphoma is different. There are over 60 different types of lymphoma and each one should be treated in slightly different ways. Every patient is different. Our experts focus only on taking care of patients with lymphoma. Your treatment for non-Hodgkin lymphoma cancer will be customized to your particular needs. One or more of the following therapies may be recommended to treat the cancer or help relieve symptoms.
Chemotherapy
Chemotherapy kills fast-growing cells, including cancer cells. This is the treatment most often used for non-Hodgkin lymphoma. And since chemotherapy may lower certain types of blood cells, a transfusion of a type of drug called blood cell growth factors may be needed. Liposomal drug delivery is an advanced way of giving chemotherapy that may help it be more effective.
Radiation Therapy
Radiation therapy uses focused beams of energy to kill cancer cells. Radiation therapy may be used in early-stage lymphoma or to help symptoms such as pain. It is seldom the only treatment given.
Proton Therapy
Proton therapy delivers high radiation doses directly to the tumor site, with minimal damage to nearby healthy tissue. For some patients, this therapy results in better cancer control with fewer side effects.
The Proton Therapy Center at UT MD Anderson is one of the largest and most advanced centers in the world. It’s the only proton therapy facility in the country located within a comprehensive cancer center. This means this cutting-edge therapy is backed by all the expertise and compassionate care for which UT MD Anderson is famous.
Immunotherapy
Instead of attacking the disease itself, immunotherapy drugs help the body fight cancer. Sometimes they have fewer side effects than other treatments.
Immunotherapy for Non-Hodgkin lymphoma may include:
- Chimeric Antigen Receptor (CAR) T cell therapy
- Monoclonal antibodies including rituximab
- Biological therapies that develop antibodies to help the body fight the cancer
- Immune modulators, such lenalidomide, that modify the environment of the tumor cell and allow the immune system to kill the cancer
- Targeted therapies that attack cancer cells by using small molecules to block pathways cells used to survive and multiply
- Small molecule treatments
Stem cell transplantation: If non-Hodgkin lymphoma does not respond to chemotherapy or if it returns, your doctor may recommend a stem cell transplant. A stem cell transplant is a procedure that replaces defective or damaged cells in patients whose normal blood cells have been affected by cancer. Also, since chemotherapy often destroys healthy cells in the blood and bone marrow, patients who have certain types of chemotherapy may need stem cell transplants.
Watchful waiting: This approach involves closely monitoring non-Hodgkin lymphoma without active treatment. Sometimes this is appropriate for some patients with low-grade lymphomas.
Learn more about non-Hodgkin lymphoma:
Learn more about non-Hodgkin lymphoma clinical trials.
Treatment at UT MD Anderson
Clinical Trials
UT MD Anderson patients have access to clinical trials offering promising new treatments that cannot be found anywhere else.
Becoming Our Patient
Get information on patient appointments, insurance and billing, and directions to and around UT MD Anderson.
Counseling
UT MD Anderson has licensed social workers to help patients and their loved ones cope with cancer.
Featured articles
myCancerConnection
Talk to someone who shares your cancer diagnosis and be matched with a survivor.
Prevention & Screening
Many cancers can be prevented with lifestyle changes and regular screening.
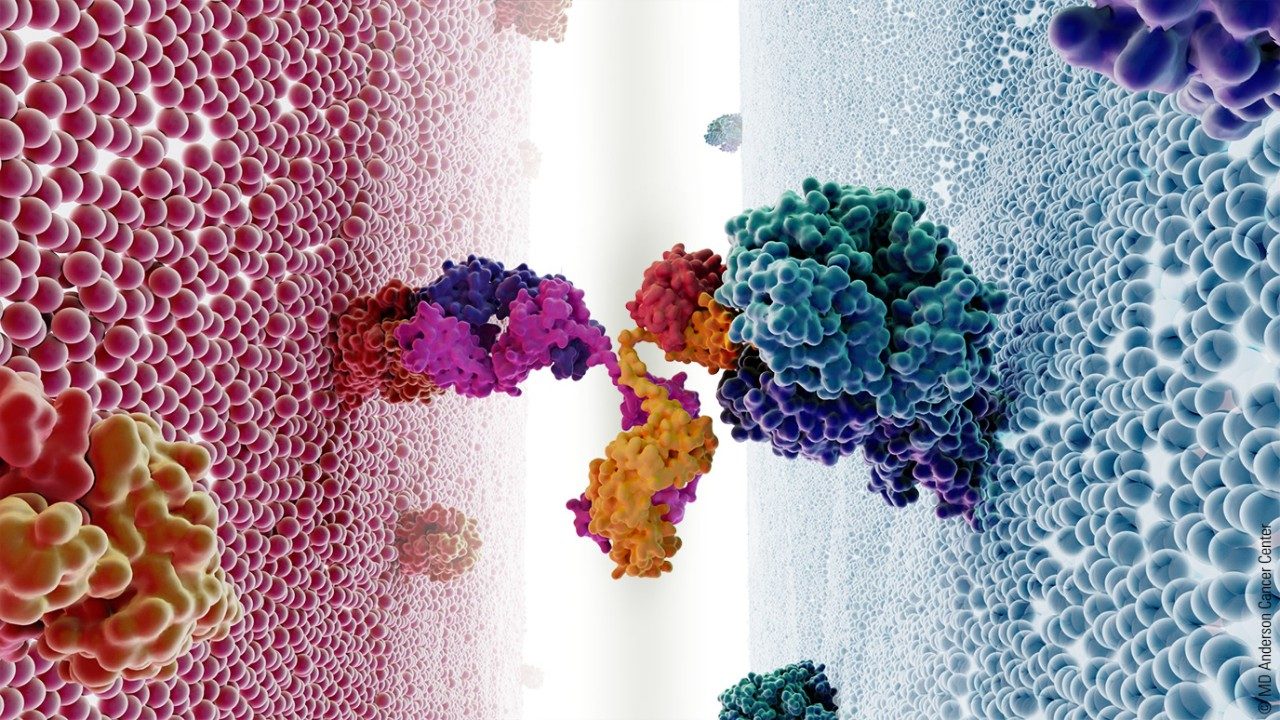
Bispecifics antibodies: The next big thing in lymphoma treatment
CD20-targeting monoclonal antibodies and immune checkpoint inhibitors started a revolution in cancer treatment by using immune cells to treat cancer. They’ve been followed by other therapies that harness immune cells to eliminate cancer, including tumor infiltrating lymphocytes (TIL) and CAR T cell therapy.
Now researchers are exploring bispecific antibodies such as mosunetuzumab and glofitamab as another way of using the body’s natural defense cells as cancer treatment. These bispecific antibodies treat cancer by engaging the tumor-specific antigen CD20 and the CD3 antigen on T cells.
"Bispecifics allows us to overcome the limitations of the immune system failing at its job: recognize and eliminate cancer cells,” says lymphoma specialist Loretta Nastoupil, M.D. “We facilitate that process by giving an antibody that's going to target the tumor and also engage and activate T cells.”
Lymphoma is primed for bispecifics
The approval of the monoclonal antibody rituximab in 1999 led to a significant improvement in overall survival in follicular lymphoma. Thanks to that advancement, Nastoupil says there’s already proof of concept that antibodies work well to treat lymphoma.
CAR T cell therapy also uses an antigen receptor that’s directed at the antigen CD19. The therapyhas shown curative results for some patients. The therapy improves a patient’s own T cells by engineering them in a laboratory with a chimeric antigen receptor, but the personalized therapy has its challenges. “It’s logistically challenging and grossly underutilized,” Nastoupil says.
Bispecific antibodies fall somewhere in the middle. Nastoupil believes they’re poised to open access to T-cell engaging therapies to more patients. “It’s off-the-shelf, and it targets an antigen that we already know is a successful target, thanks to rituximab,” she says.
CD20 is present on all malignant B cells, making for an attractive target
In addition to being more accessible as an off-the-shelf product, findings from early clinical trials show promise with CD20-targeting bispecifics in lymphoma.
“It’s a great target because it’s only expressed by mature or malignant B cells,” Nastoupil says. The lymphoma cells are easily targeted, and eradication of mature B cells doesn’t cause harm.
That means fewer and less severe side effects for patients. Even if a bispecific eliminates mature B cells, it will result in a temporary drop in antibody levels, Nastoupil says. But they recover with time. “Although patients are at a higher risk of infections like sinusitis and bronchitis, in general most are not life-threatening,” Nastoupil says.
As with other cellular therapies, bispecifics can cause a unique side effect called cytokine release syndrome, which can be life threatening. Patients experience fever, chills and flu-like symptoms. Several tactics are being explored to reduce the risk of cytokine release syndrome, Nastoupil says. One method is called dose step-up or dose ramp-up. The dosage of the bispecific is slowly increased to control T cell engagement and the cytokine reaction. Researchers are also exploring subcutaneous delivery. Instead of being administered through the veins, the drug is injected under the skin. “It’s resulting in slower, steadier absorption,” Nastoupil says.
Patients can also experience neutropenia and fatigue, but Nastoupil says they’re common for patients receiving a third line of treatment or later for lymphoma.
“The side effects of bispecifics aren’t new, and the rates of serious events are quite low,” Nastoupil says. That holds particularly true when considering the toxicity profile of bispecifics with other targeted therapies such as PI3 kinase inhibitors, CAR T cell therapy and multi-agent chemotherapy.
Bispecifics in earlier lines of therapy may have better results
Currently, bispecifics have been investigated as a third-line therapy or later. “It’s hard to know if the efficacy is better than anything out there right now because it’s still early, but we’re seeing exciting results,” Nastoupil says.
Based on bispecifics’ safety profile and promising signs of efficacy, the next step in advancing these drugs is to test them in patients in earlier settings. “Since we’re seeing bispecifics work well as third-line therapy or later, we expect them to do even better in earlier lines,” Nastoupil says.
Combinations in earlier settings could improve efficacy
There are also opportunities to improve efficacy through combinations with other drugs. “We want to build on them with either chemotherapy in large cell lymphoma or with immunotherapy in follicular lymphoma,” Nastoupil says.
For follicular lymphoma, mosunetuzumab plus lenalidomide is being explored in a randomized Phase III study. As an oral immune modulator, lenalidomide works by enhancing the ability of T cells and natural killer cells to clear cancer cells, and the bispecific mosunetuzumab helps direct the cells to and into the tumor microenvironment. They work in synergy. “The cells have direction, and they’re activated,” Nastoupil says. “Now, they’re seeing what they should be removing and doing their job.”
In large cell lymphoma, mosunetuzumab or glofitamab has been combined with polatuzumab, a CD79B antibody drug conjugate. “These combinations work like chemoimmunotherapy,” Nastoupil says. Polatuzumab delivers targeted chemotherapy to the tumor and the bispecific pulls in the immune cells.
These combinations have been tested in the third-line setting, but larger studies in the earlier settings are now underway, Nastoupil says.
Defining sequencing for use in the community
Nastoupil anticipates several bispecifics will be approved by the Food and Drug Administration (FDA) soon. As they enter the treatment landscape, she says the next challenges are to determine when they should be used instead of other therapies and in what sequence. Nastoupil thinks it will be center-specific because of the low, but real, risk of side effects.
“Ensuring these drugs can be safely delivered to patients in the community it critical for access,” Nastoupil says. While the risk of a severe cytokine reaction is low, those patients need to be admitted to a hospital when that occurs. “Anything that can be done to reduce that risk of hospitalization is being explored right now,” Nastoupil says. “These drugs could change how lymphoma is treated, but we have to make sure it’s safe for all.”
Request an appointment at MD Anderson online or by calling 1-877-632-6789.
Help #EndCancer
Give Now
Donate Blood
Our patients depend on blood and platelet donations.
Shop UT MD Anderson
Show your support for our mission through branded merchandise.