request an appointment online.
- Diagnosis & Treatment
- Cancer Types
- Multiple Myeloma
Get details about our clinical trials that are currently enrolling patients.
View Clinical TrialsMultiple myeloma, also known as Kahler's disease, is an uncommon blood cancer that affects the bone marrow, the body's blood-forming system, and can lead to kidney problems.
Multiple myeloma, also known as Kahler's disease, is an uncommon blood cancer that affects the bone marrow, the body's blood-forming system, and can lead to kidney problems.
What is Multiple Myeloma?
Multiple myeloma is a cancer of the plasma cell. Normal plasma cells are a type of white blood cell that helps make up your immune system. They are located within the bone marrow - the spongy interior of bones that produces blood cells. When your body is fighting an infection, plasma cells produce antibodies (proteins) which attack viruses and bacteria.
If a plasma cell becomes cancerous, it multiplies rapidly. This is multiple myeloma. The malignant plasma cells may crowd out normal blood-forming cells within the bone marrow, reducing the production of healthy blood cells. Additionally, rather than producing infection-fighting antibodies, the cancer cells begin to produce an abnormal antibody called a monoclonal protein (m protein) or paraproteins. In the urine, they are called Bence Jones proteins. These proteins do not fight against infection.
Symptoms
Multiple myeloma often does not have early symptoms. This can make it difficult to diagnose in the beginning stages. Symptoms of multiple myeloma may include:
- Fractures: Myeloma cells trigger the destruction of the surrounding bone. The weakened area of bone is more likely to break. This is called a pathological fracture.
- Bone pain, especially in the middle and/or lower back, rib cage or hips. The pain can be mild or severe depending on the extent of the multiple myeloma, the speed with which it has developed, and whether fracture or nerve compression has occurred.
- Fatigue and/or shortness of breath: Myeloma can cause anemia, which can lead to feeling short of breath with exertion or tiredness more than usual.
- Confusion: Multiple myeloma can lead to high calcium levels in the blood and/or kidney failure. This can lead to confusion. Confusion can also be related to hyperviscocity (overly thick) blood.
- Numbness or weakness: Multiple myeloma is sometimes associated with nerve compressions, which can lead to numbness in the limbs. Hyperviscocity can also lead to weakness or numbness of the face, arms, or legs
- Leg swelling: Multiple myeloma damages the kidneys, preventing them from working effectively. This means your body can't get rid of extra salts and fluids, which can produce swelling
- Appetite changes: High calcium levels in the blood and/or kidney failure can also cause a decrease in appetite, weight loss and nausea.
- Frequent infection: Because myeloma cells crowd out normal white blood cells, which fight infection, there is a risk of infection. Common myeloma infections include pneumonia, bladder or kidney infections, sinusitis and skin infections.
- Excessive thirst due to high blood calcium levels and kidney damage.
Complications
As a blood cancer, multiple myeloma interacts with many other body systems and can lead to a variety of issues. Complications of multiple myeloma include:
- Anemia: A shortage of red blood cells, caused when abnormal plasma cells crowd out healthy blood-producing bone marrow cells
- Leukopenia: A shortage of white blood cells, which can compromise your immune system
- Bone erosion or bone lesions: Myeloma cells produce substances called cytokines, which can trigger bone cells (osteoclasts) to destroy surrounding bone. When more than 30% of the bone has been destroyed, x-rays show a thinning of the bone (osteoporosis) or dark holes (lytic lesions). The weakened area of bone is more likely to break.
- High blood calcium levels: Related to bone erosion. Calcium stored in bones enters the bloodstream, which impacts other body systems.
- Hyperviscosity: In some cases, large amounts of m proteins can make your blood thicker, which impedes flow into smaller blood vessels
These complications are all associated with different signs and symptoms.
Causes
In 2015, an estimated 124,733 people in the United States were living with this disease. The exact cause of multiple myeloma is not known and no avoidable risk factors have been found. However, certain things appear to make you more likely to develop the disease.
Risk factors for multiple myeloma:
- Age: Over 65
- Gender: Men are slightly more likely to develop myeloma.
- Race: Black people are twice as likely to develop myeloma than white people.
- Radiation exposure
- Family history: If a parent, brother or sister has the disease, your risk is four times higher. However, this is rare.
- Working in oil-related industry: While some studies suggest this, it has not been proven.
- Obesity
- Other plasma cell disorders: If you have had one of the following you are at higher risk:
- A precancerous condition called monoclonal gammopathy of undetermined significance (MGUS)
- A single tumor of plasma cells (solitary plasmacytoma)
- Smoldering myeloma, a precancerous condition that affects the plasma cells and can turn into multiple myeloma
Learn more:
Multiple Myeloma: Blood Cancer in Bone Marrow news
UT MD Anderson is #1 in Cancer Care
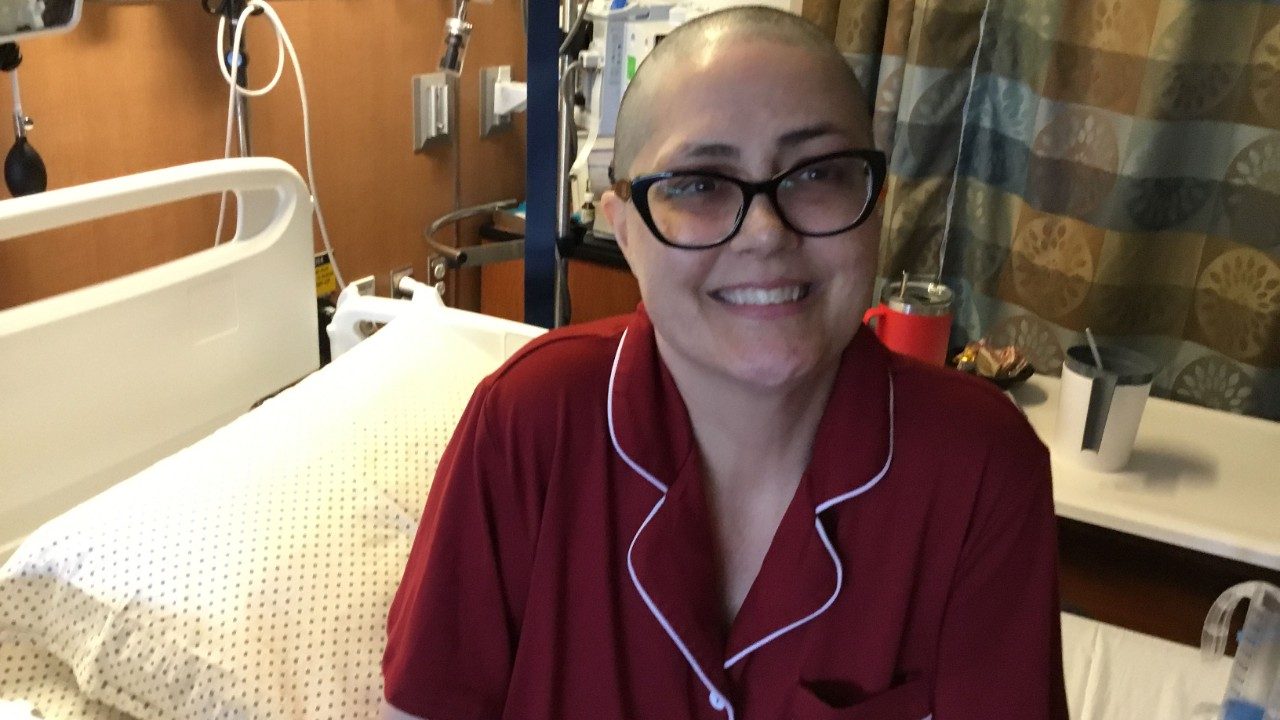
Multiple myeloma and spine cancer survivor: ‘Always think about a second opinion’
In 2022, Emily Shults, then 47, had recently moved to Magnolia, Texas, from California, to be closer to family. During the move, she remembers dealing with bad back pain. Emily wasn’t too concerned. “I thought I’d tweaked something in the move,” she says.
But she went to a local doctor north of Houston to get checked out.
Back pain leads to cancer diagnosis
Blood work showed she was borderline anemic. She was referred to physical therapy. Emily recalls telling the physical therapist, “You’ll think I’m insane, but the pain is no longer in my shoulder; it’s in my lower back and radiating down into my leg.”
Throughout the autumn of 2022, her pain got worse. On a trip, the pain was so bad she had trouble sitting in a car. “I thought I was going to keel over!” she says. Emily’s legs felt numb as she stood up a few days later. As she started to fall, she put her arm out and then heard her arm snap. A call to 911 led her to the emergency room at a local hospital.
Emily had broken her arm. Scans revealed a lesion on her humerus, the large bone of the upper arm. But that wasn’t all: “My spine was caving in on itself because of lesions, and I had a lesion on my left pelvis and left rib,” recalls Emily.
She was diagnosed with multiple myeloma, a blood cancer that affects the bone marrow and weakens the bones. “The rug was pulled out from us,” Emily says. “I was like, ‘I have back pain! What do you mean, I have cancer?’”
Second opinion at MD Anderson changes treatment plan
Emily started radiation therapy at her local hospital, but she remained concerned that she needed more care. “Everyone said, ‘You need a myeloma specialist,” she recalls. “’ Go to MD Anderson.’”
She scheduled an appointment at MD Anderson.
But a week before her MD Anderson appointment, Emily’s legs felt heavy. Her doctor recommended she go to MD Anderson’s Acute Cancer Care Center right away. There, MD Anderson doctors found clots in her legs and a pulmonary embolism.
Emily spent a week in the hospital at MD Anderson while her care team developed a treatment plan. First, she would need chemotherapy. Her doctors recommended a combination medication, carfizomib lenalidomide dexamethasone (KRd), widely used for multiple myeloma. She also would receive daratumumab, a targeted therapy with monoclonal antibodies.
The goal was for Emily to have no minimal residual disease (MRD), meaning no trace of the disease could be found.
She met with Claudio Tatsui, M.D., a neurosurgeon who specializes in spine surgery, to explore options for treating the damage on her spine from the cancerous lesions. Tatsui felt she wasn’t stable enough for surgery yet, so she began physical therapy nearby.
Spinal surgery and stem cell transplant help multiple myeloma patient regain independence
A few months later, Emily had no minimal residual disease after successful chemotherapy. Her doctors felt she was ready for spinal stabilization surgery to help her walk and independently carry out daily activities again.
“It was very scary, but I think Dr. Tatsui and his team did everything they could to make me feel comfortable,” says Emily of the June 2023 surgery. “He was very thoughtful and careful.” Tatsui and his team explained every detail of the surgery as well as potential side effects. “They did everything they could to make sure I understood,” Emily says.
The surgery was a success. In addition, her MD Anderson doctors recommended a stem cell transplant, which replaces diseased bone marrow with healthy bone marrow cells.
Two months after her spine surgery, Emily had a stem cell transplant under the care of stem cell transplant specialist Muzaffar Qazilbash, M.D. “I went in the hospital using a walker, had surgery, and in July of 2023, I did my stem cell and went in with a rollator,” reflects Emily on her progress.
Today, Emily remains MRD-negative. Effectively, she is in remission. While she continues maintenance chemotherapy, she is back to walking and doing everything on her own. Her broken arm bone has also fully healed.
How does Emily feel about her time at MD Anderson? “Everyone has been really good about listening to me,” she says. “Every time I’ve had a challenge, they listen and connect me with specialists and rule things out.” For instance, a few months after her stem cell transplant, Emily had fluid buildup in her lower legs and feet. It was edema. This condition can occur in multiple myeloma patients after a stem cell transplant due to the cumulative effects of chemotherapy, white blood cell growth factors, and nutrition changes. “My legs felt like cement blocks,” Emily recalls.
Her care team responded immediately, putting her on a medication called furosemide twice a day and giving her a referral for therapy near her home. “Patients with severe edema can benefit by getting care at a lymphedema clinic or center,” says Qazilbash. “It is fully reversible in almost all cases.”
Emily’s team also ordered compression stockings for her. Today, the swelling is much more manageable.
Staying positive and engaged in care
Reflecting on her diagnosis and surgery, Emily says, “I allowed myself those days to cry, but focusing on the positive was a big driver for me.” Even on her worst days, she’d look for the positive. “As we’d see the doctors and nurses, they said, ‘You have a bright light,’” she recalls.
Being an active participant in her treatment and recovery helped Emily cope and heal. “Stay focused on the prize, partner with your team and figure out how to get through the setbacks,” she tells other patients.
Asking questions helped Emily feel empowered during her treatment, and she encourages other patients to do the same: “Make sure you are your own advocate; ask questions because that gives you peace of mind,” says Emily, who uses MyChart for questions between appointments. “If I can’t talk to the doctor on the phone, someone on their team is responding.”
Staying connected to loved ones also helped Emily and her family throughout her treatment. Her husband used social media to keep friends and family updated. “It’s hard on your caregivers; he needed an outlet,” she says.
Reflecting on her journey, Emily offers one last suggestion for other patients. “Always think about a second opinion. And don’t be afraid to say you need help or support.”
Request an appointment at MD Anderson online or call 1-877-632-6789.

Multiple myeloma: When to treat and when to wait
Multiple myeloma is a cancer that develops in a type of white blood cell called plasma cells. Normally, these cells help us make antibodies to fight infection. But when something goes wrong during cell division, they can live forever.
Some patients live with multiple myeloma for years without any negative effects. This is called smoldering myeloma. But eventually the myeloma cells can make things go haywire in the body, affecting patients’ red blood cell counts, the kidneys, calcium levels and even the bones.
To understand how multiple myeloma affects the body and when it’s best to treat it, we talked with Krina Patel, M.D.
How does multiple myeloma affect the body?
One common issue related to the myeloma cells is anemia. Often the cancerous cells take up so much space in the bone marrow that they push the red blood cells out.
Myeloma can also damage the kidneys. It can cause inflammation and block the filters of the kidney, so some patients may notice foam or bubbles in their urine.
Some patients also experience bone issues. Myeloma can cause lytic lesions, which are like holes in the bones. The myeloma cells can also cluster in or just outside the bone and form tumors called plasmacytomas. The cancer accelerates osteoporosis as well by helping the cells that break down bone and slowing the cells that build new bone.
Although it's rare, the tumor-like plasmacytoma can also occur elsewhere in the body, like the lungs or the liver, but that typically occurs with more advanced disease. This is called extramedullary disease.
Most patients don't experience all these issues – usually one or two.
Why does timing matter when treating multiple myeloma?
Myeloma isn't curable yet, but it's very treatable. The key is deciding when to treat it to manage the symptoms. Our goal is to improve the patient’s quantity of life without compromising quality of life.
Anemia can cause fatigue. Bone issues can cause fractures, so it’s harder to walk. Kidney failure will require you to go on dialysis. All of this impacts quality of life. But if we treat the disease too early, it can become resistant to treatment and ultimately decrease your lifespan.
Our goal is to put the myeloma into hibernation for as long as we can. And then we will treat you again when we think that it's about to cause more problems.
How is multiple myeloma initially treated?
75% of patients have standard-risk disease, which is myeloma that we think will respond to treatment well. Although radiation therapy and surgery are used in some rare situations, patients typically receive three or four rounds of chemotherapy. However, the chemotherapy we use to treat multiple myeloma is different than what’s used for other cancers. The side effects aren’t as severe, and we try to limit the impact on your daily routine. Patients may come into the clinic once a week for a 30-minute chemotherapy infusion or a shot, and then they are likely taking chemotherapy pills at home daily for a few weeks.
In addition, patients receive steroids to help with the inflammation in the bones and the kidneys. This can also help reduce pain. More importantly, steroids kill the myeloma cells.
Through blood draws, we monitor protein levels to see how the myeloma is responding to treatment. Once the myeloma protein level is down, patients typically receive a stem cell transplant. This allows us to give a big dose of chemotherapy that wipes out the bone marrow and puts the myeloma into deeper, longer hibernation.
After that, patients go on chemotherapy maintenance. Standard-risk patients receive a lower dose chemotherapy pill until the myeloma protein numbers start to rise again, which is on average about four to five years. Patients with high-risk disease have a more aggressive maintenance plan that includes a combination of chemotherapies. They typically have about two to three years until the disease flairs up again.
What happens when the myeloma returns?
Multiple myeloma is smart. Each time it comes back, it’s a bit more aggressive, but the treatment plan is similar. After using one drug as the first-line treatment, the myeloma typically learns to go around it. But studies have shown that when we use a combination of drugs (typically three) to treat relapsed disease, it tends to stay down longer.
There is a lot of research being done in myeloma, so we have new drug options that we can try when chemotherapy fails. We’re seeing an explosion of immune therapies like CAR T cell therapy, monoclonal antibodies and targeted therapies. But because we have so many options for treatment, it’s more challenging to select the right approach for each patient.
If you’ve recently been diagnosed with myeloma, I suggest you seek care in as specialty center like MD Anderson so that you have the best drugs for you, sequenced in the best order for your disease. That way, we can give you as many years possible to enjoy life without letting myeloma disrupt it.
Request an appointment at MD Anderson online or by calling 1-877-632-6789.
Why choose UT MD Anderson for your multiple myeloma treatment?
Treatment for multiple myeloma may continue for extended periods, with repeated remissions and recurrences. With so many new treatment options and combinations available, studies have shown improvements in clinical outcomes for patients who have a relationship with an oncologist specializing in the treatment of myeloma. For this reason, our teams of experts and specially-trained support professionals forge close medical relationships with you to be sure you receive personal, customized care. This individualized attention gives you the best chance of a full and healthy life.
Pioneering Advances
UT MD Anderson has been instrumental in some of the biggest advances in multiple myeloma treatment, including Revlimid® (lenalidomide), a derivative of thalidomide. It is one of the most important discoveries in myeloma therapy. We've also found that combining this drug with chemotherapy drugs often has a profound effect, even after other treatments have failed.
At UT MD Anderson, you benefit from one of the most active research programs in the United States, which includes a prestigious federally funded SPORE (Specialized Program of Research Excellence) program. This means we are able to offer a wide range of clinical trials (research studies) for every stage of multiple myeloma.
Cancer, after all, is a battle, and a positive attitude and hope are crucial weapons. So far, I win!
Jennifer Davis
Survivor
Treatment of Multiple Myeloma
Featured Articles
Your questions about BCMA and multiple myeloma, answered

CAR T cell therapy puts smoldering and multiple myeloma survivor in remission

10-year multiple myeloma survivor: Why I joined a clinical trial at MD Anderson

Medical laboratory scientist faces her own multiple myeloma diagnosis
5 questions about multiple myeloma, answered
6 facts about smoldering myeloma

Multiple myeloma survivor and employee: ‘MD Anderson really is the best’
Multiple myeloma survivor: Why I chose MD Anderson for my cancer treatment
Clinical Trials
UT MD Anderson patients have access to clinical trials offering promising new treatments that cannot be found anywhere else.
Becoming Our Patient
Get information on patient appointments, insurance and billing, and directions to and around UT MD Anderson.
myCancerConnection
Talk to someone who shares your cancer diagnosis and be matched with a survivor.
Prevention & Screening
Many cancers can be prevented with lifestyle changes and regular screening.
Counseling
UT MD Anderson has licensed social workers to help patients and their loved ones cope with cancer.
Help #EndCancer
Give Now
Donate Blood
Our patients depend on blood and platelet donations.
Shop UT MD Anderson
Show your support for our mission through branded merchandise.
