Researchers identify Rb1 as a predictive biomarker for a new therapeutic strategy in some breast cancers
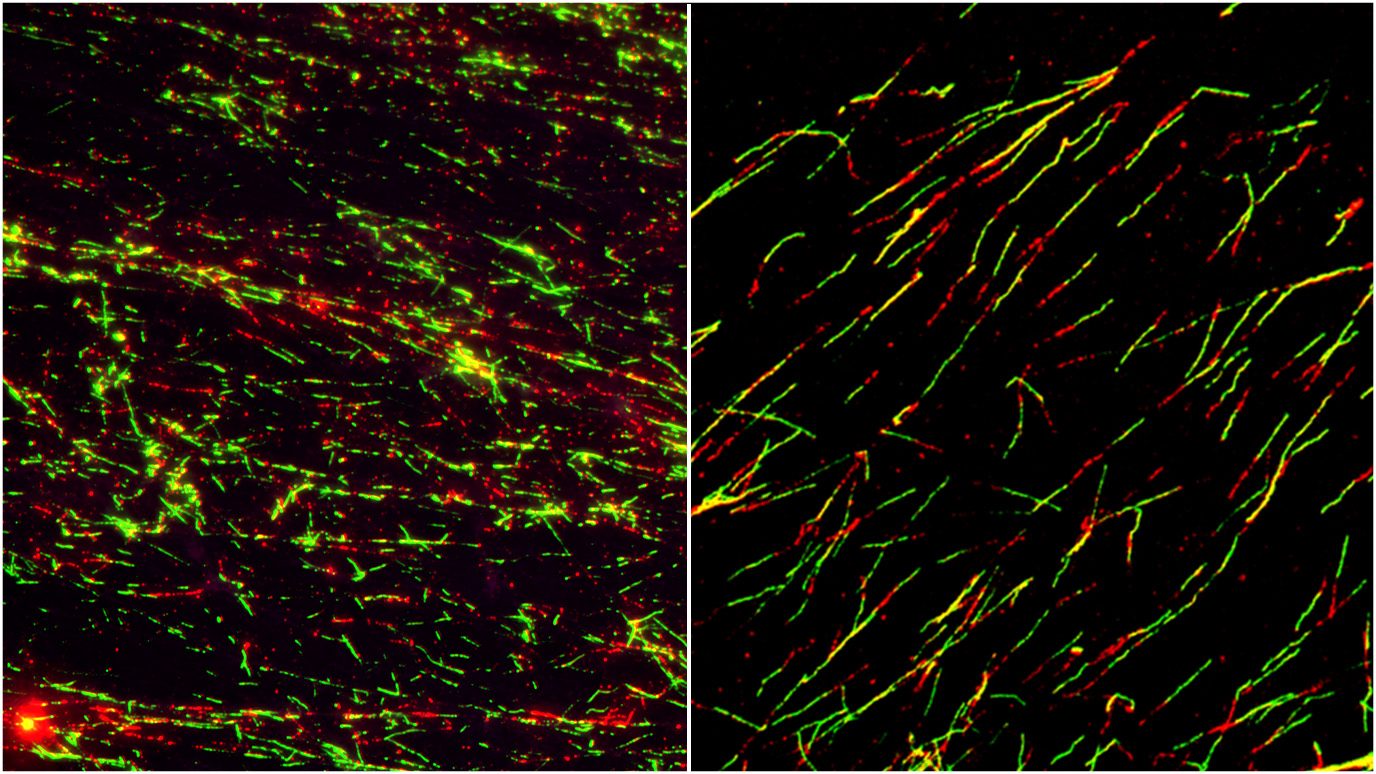
Novel targeted therapy regimens show promise in aggressive AML subtype
MECOM-rearranged acute myeloid leukemia (AML) is an aggressive and hard-to-treat leukemia
Researchers combined three inhibitors...

Promising target improves antitumor response in preclinical models of pancreatic cancer
Inhibiting GFER along with...
Proton therapy shows survival benefit in Phase III trial for patients with head and neck cancers
A new study published today in...

SABCS 2025: Diagnostic breast MRI may be unnecessary for some patients with early-stage breast cancer
Study finds pre-surgical MRI does not improve five-year rates of local regional free survival over mammograms alone in patients with stage...

ASH 2025: New data highlights promise of pivekimab sunirine in two aggressive blood cancers
Two clinical studies demonstrate high response rates by patients with two hard-to-treat and aggressive blood cancers
Pivekimab...

ASH 2025: Researchers identify CD40 as potential biomarker for a type of nodal T cell lymphoma
Researchers characterized the microenvironment of a nodal T cell lymphoma that is difficult to diagnose and treat
The study...

ASH 2025: Shorter azacitidine regimen safely improves outcomes in lower-risk MDS patients
Azacitidine is a type of targeted therapy known as a hypomethylating agent, commonly used in treating myelodysplastic syndromes (MDS)...
ASH 2025: Results show long-lasting benefits of CAR T cell therapy for hard-to-treat lymphoma
Lisocabtagene maraleucel (liso-cel), a CAR T cell therapy, achieved an overall response rate of 97% in the TRANSCEND FL study
Durable...

ASH 2025: New MD Anderson-developed antibody boosts immune response against blood cancers and solid cancers
New investigational therapy 77A is an antibody that converts a cancer survival protein (HSP70) into an immune system trigger
Targeting...
ASH 2025: CAR T cell therapy shows promising Phase II trial results in multiple myeloma
Anito-cel is an experimental CAR T cell therapy for relapsed/refractory multiple myeloma patients
In updated Phase II trial results...
Researchers identify target to overcome treatment resistance in preclinical models of KRAS-mutant cancers

