Research Directions
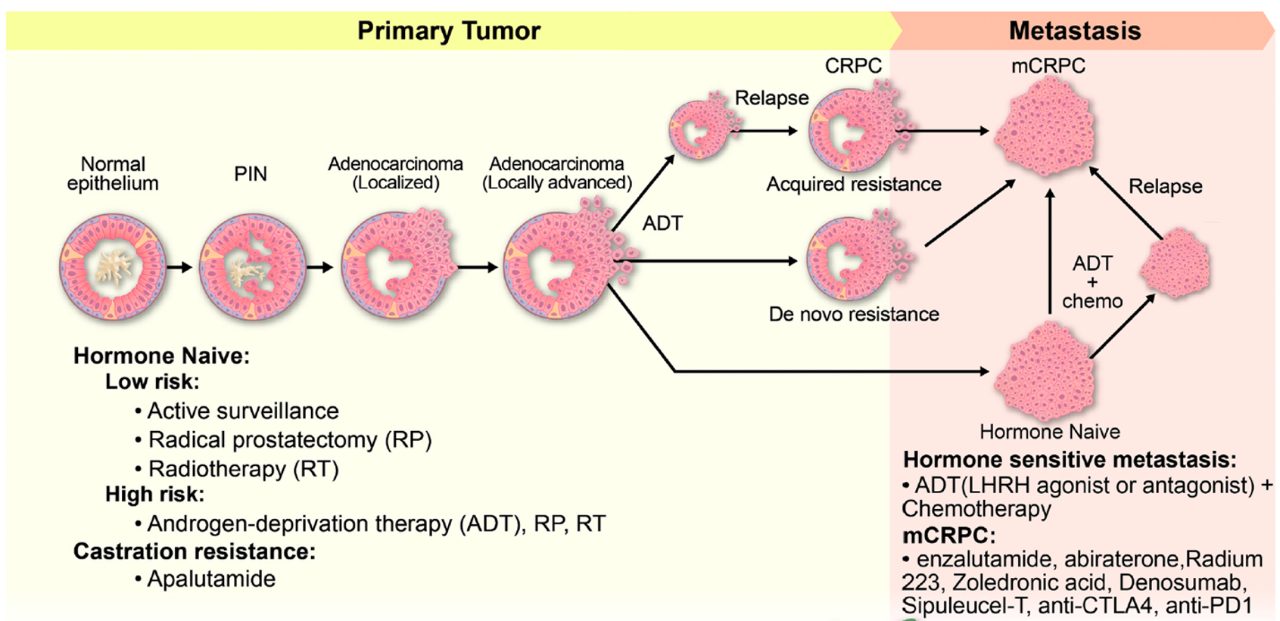
Prostate cancer is the most commonly diagnosed cancer and the leading cause of cancer death in men worldwide. Although nearly all patients respond to androgen deprivation therapy, most patients progress to castration-resistant prostate cancer with high rates of metastasis and mortality. Over the past decade, large-scale cancer genome studies have uncovered emerging genetic alterations in advanced prostate cancer. It is vital to identify and characterize genetic determinants that drive prostate cancer development, metastatic progression and resistance to therapy. The Zhao Laboratory focuses on prostate cancer genetics and biology, with a special interest in the crosstalk between cancer cells and immune components in the tumor microenvironment and metastatic niche. Our major research directions include:
- Identifying the genetic events that drive prostate cancer development, metastatic progression, and therapy resistance.
- Characterizing cancer-cell intrinsic genetic determinants that reshape the tumor microenvironment and metastatic niche.
- Developing biomarker-driven immunotherapies and combinatorial strategies for precision cancer medicine.
Identifying the genetic events that drive prostate cancer development, metastatic progression and therapy resistance.
Genetically engineered mouse models (GEMMs) produce spontaneous tumors in immunocompetent mice, offering unique systems for studying tumor development, metastatic progression and tumor-host interactions. Our team utilizes GEMMs to investigate the role of emerging tumor suppressor genes and oncogenes in prostate cancer progression and response to therapies. Our laboratory has established and characterized several GEMMs to mimic different genetic subtypes of prostate cancer, including PTEN, TP53, SMAD4, CHD1 and SPOP. Combined with high-throughput multi-omics technology and in-depth mechanistic studies, these GEMMs enable us to uncover novel functions of hotspot genetic events, alone or in combination, during different stages of prostate cancer. Additionally, we generate 3D tumor organoids and syngeneic prostate cancer models from these GEMMs, providing unique tools for studying tumorigenesis, metastasis, cancer cell-immune cell interactions and drug response. These studies facilitate the identification of therapeutic vulnerabilities for different molecular subtypes of prostate cancer (Cancer Discovery 2020; Cancer Research 2022; Science Translational Medicine 2023; Nature Cancer 2025; Nature Communications 2025).
Characterizing cancer-cell intrinsic genetic determinants that reshape the tumor microenvironment and metastatic niche.
Immunosuppressive myeloid cells, including tumor-associated macrophages (TAMs) and myeloid-derived suppressor cells (MDSCs), play a crucial role in cancer progression and the development of therapy resistance. Understanding the crosstalk between cancer cells and myeloid components in the tumor microenvironment and metastatic niche advances our knowledge of tumor-host interactions and facilitates the development of effective therapeutics for advanced prostate cancer. By integrating our state-of-the-art mouse modeling systems with single-cell-based immunoprofiling approaches, we aim to elucidate how genetic alterations in cancer cells reprogram the abundance, phenotype, and functions of myeloid cells in prostate tumors and metastatic diseases (Cancer Discovery 2020; Science Translational Medicine 2023; Nature Communications 2025).
Developing biomarker-driven immunotherapies and combinatorial strategies for precision cancer medicine
Immunotherapy only showed modest anti-tumor activities in advanced prostate cancer, partially due to a lack of T cell infiltration, low mutational burden and presence of suppressive immunocytes. Targeting the immunosuppressive components in the tumor microenvironment and metastatic niche presents significant opportunities to improve immunotherapy. Our goals are to explore these combinatorial strategies and identify the molecular subtypes that respond better to these therapies. By elucidating in-depth mechanisms and conducting preclinical trials, we aim to develop biomarker-driven combinatorial immunotherapy for precision prostate cancer medicine (Cancer Discovery 2020; Science Translational Medicine 2023; Nature Communications 2025). B7-H3 is an immune checkpoint and an emerging target for immunotherapy. We are particularly interested in understanding the biology of B7-H3 signaling, elucidating its impact on the tumor microenvironment and designing personalized and combinatorial treatments to enhance the efficacy of B7-H3-targeted therapies in advanced prostate cancer.
Give Now
Research Areas
Find out about the four types of research taking place at UT MD Anderson.
