Research
The Wang Lab focusses on the crosstalk between cancer cells and the tumor microenvironment (TME) and its influences on cancer progression, metastasis and therapy resistance at the molecular and cellular levels. We use a multi-disciplinary approach to define how epigenetic and metabolic programs are rewired in cancer and how we can effectively target these aberrant epigenetic and metabolic pathways to treat human cancer. This work spans the areas of epigenetics, cancer metabolism, the tumor microenvironment and immunometabolism using novel genetic mouse models, proteomics, epigenomics, metabolomics and cutting-edge single cell technology. Ultimately, we aim to translate our findings into novel targeted therapies for cancer.
Current Projects
Our current work largely focuses on four major areas:
- The role of myeloid-derived suppressor cells in cancer progression, metastasis, and therapeutic resistance
Previously, we demonstrated that targeting the Yap1-dependent infiltration of myeloid-derived suppressor cells (MDSCs) into tumors can be an effective therapeutic approach in advanced prostate cancer. Part of our ongoing work is to (a) investigate the role of MDSCs in the development of castration-resistant prostate cancer, (b) elucidate the molecular mechanisms by which MDSCs suppress anti-tumor immunity, (c) identify novel therapeutic targets in MDSCs and develop inhibitors and (d) identify effective combination therapy using MDSCs targeting agent and immune checkpoint inhibitors.
- The role of epigenetic regulators in driving cancer progression cells, metastasis, and therapeutic resistance, with a focus on histone lysine demethylases, histone lysine methyltransferases, and chromatin remodelers
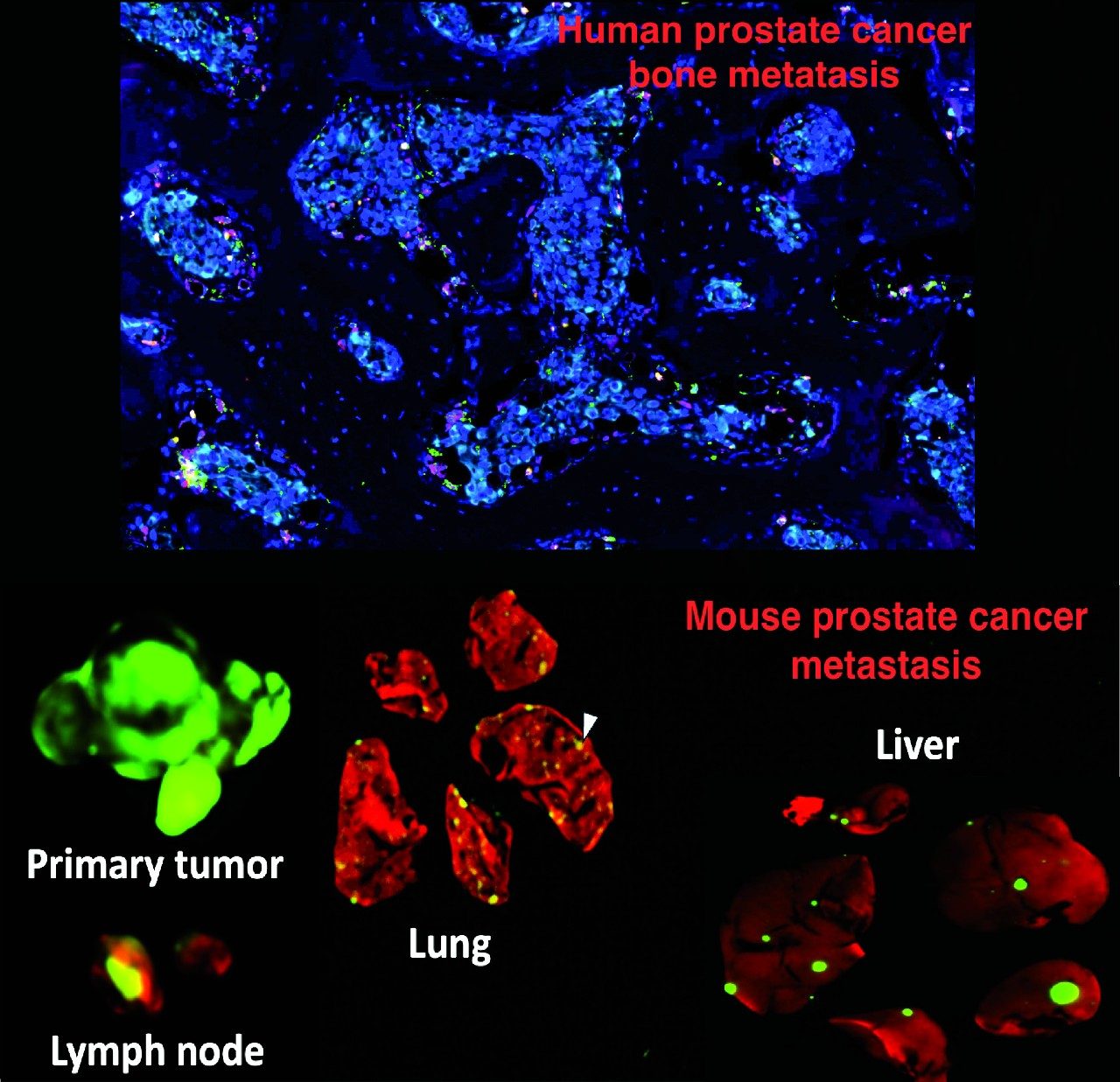
We recently identified histone lysine demethylase KDM4A as an epigenetic driver of tumor progression in neuroendocrine prostate cancer (NEPC), one of the most lethal subtypes of advanced prostate cancer that lacks effective therapy. We continue to study the functions of KDM4A in NEPC and examine the efficacy of KDM4A targeting agents as monotherapy and combination therapy. Also, we investigate how the epigenetic programs drive prostate cancer metastasis to bone and liver, the major metastatic sites for prostate adenocarcinoma and NEPC.
- The role of immunometabolism dysregulation in cancer progression, metastasis, and therapeutic resistance
Immunometabolism refers to the intricate interplay between immune and metabolic pathways that plays a crucial role in health and disease, including cancer. Recent studies have shown that metabolic reprogramming, a hallmark of cancer, results in metabolic stress in T cells, leading to a cold immune tumor microenvironment (TME) that severely limits the efficacy of T cell–based immunotherapy. Thus, targeting dysregulated metabolic pathways has emerged as a promising therapeutic strategy in cancer, potentially enhancing the response of patients to immunotherapy, including patients with prostate cancer (PCa). We will focus on metabolic reprogramming in neuroendocrine prostate cancer (NEPC) and MDSCs.
- Development of clinically relevant genetic mouse models for upper tract urothelial cancer (UTUC) and the study of molecular and cellular mechanisms driving UTUC progression
We are developing genetic mouse models harboring genetic aberrations commonly seen in human UTUC. We will use our UTUC genetic mouse models and human UTUC cell lines/organoids to identify driver oncogenes/tumor suppressor genes in UTUC progression and therapeutic resistance.
Give Now
Research Areas
Find out about the four types of research taking place at UT MD Anderson.
