Research
Research in the Arur Laboratory uses several different model systems to investigate the cellular principles of female fertility, and how this impacts tumor progression and metastasis. Our current research projects focus on:
- The role of maternal nutrition and environmental cues in regulating oocyte growth, development and quality
- The genetic and molecular architecture controlled by insulin signaling and maternal nutrition to regulate meiosis I
- Signal mediated control of small RNA biogenesis enzymes to coordinate oocyte to embryo transition
- Phosphorylated nuclear Dicer as a driver of aggressive tumor progression and metastasis
Role of maternal nutrition and environmental cues in regulating oocyte growth, development and quality
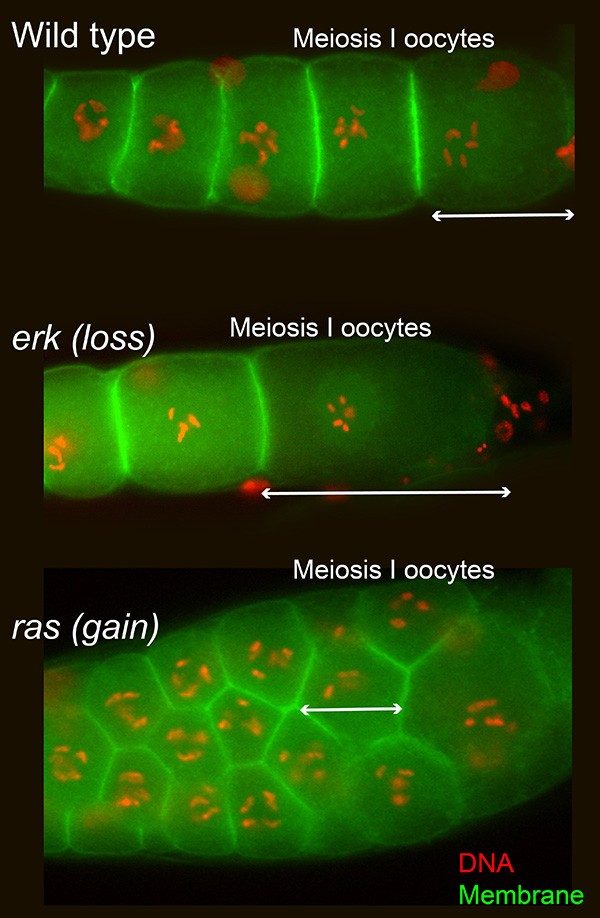
Developmental birth defects, and failed pregnancies, are often linked directly to poor oocyte quality. For example, a poor-quality oocyte with wrong number of chromosomes results in severe aneuploidy in the fetus during early development leading to embryonic birth defects or death. However, several fetal abnormalities occur despite a normal oocyte chromosomal complement, suggesting that factors in addition to chromosomal numbers regulate oocyte quality. These factors remain unknown because the cell-intrinsic molecular cues, and the cellular and physiological parameters that determine oocyte quality remain mostly uncharacterized. The growth of the oocyte to its final size and accumulation of the cytoplasmic and maternal components – critical for early fetal development – occur during meiosis I of oogenesis. Mammalian meiosis I is largely completed in the fetal ovary. Thus, deciphering the molecular cues, and physiological states within the meiosis I oocyte in vivo remains a challenge.
Caenorhabditis elegans forms an excellent model system to understand the cellular and genetic mechanisms of oocyte quality at meiosis I; meiosis I of oogenesis occurs during adulthood in C. elegans. As in vertebrates, oocyte growth and development is controlled by the ERK signaling pathway in worms (Figure). We have shown that loss of ERK activity from oocytes, results in formation of giant oocytes (Figure, middle), and conversely, excessive ERK activity results in multiple small oocytes (Figure, bottom). Both large and small oocytes result in embryonic death or infertility. We have also shown that insulin like receptor (DAF-2) and maternal nutrition regulate the RAS-ERK signaling pathway to control oocyte growth and quality.
Thus, a major goal in the lab is to investigate the cell-intrinsic determinants of oocyte quality in vivo using C. elegans, which integrates maternal nutrition-dependent signals with their control on oocyte growth and quality. We combine developmental genetics, with high resolution imaging, genomics and proteomics to uncover the mechanisms driving oocyte growth and size to dictate oocyte quality.
Investigate genetic and molecular architecture controlled by insulin signaling and maternal nutrition to regulate meiosis
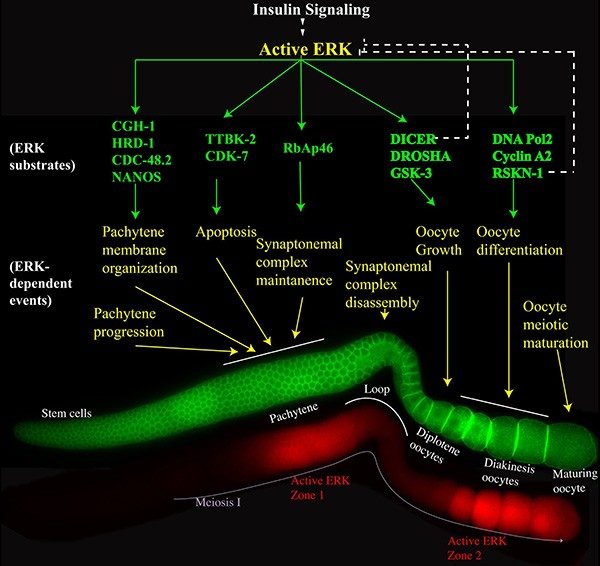
Effective communication is the hallmark of a civil society; as is effective communication among and within cells for normal development. Breakdown in this communication results in a myriad of diseases, such as birth defects and cancers. Tissues and cells use the language of cell-signaling for regulated effective cellular communication.
Our goal is to understand one such language, that used to respond to and relay extracellular signals intracellularly by the RAS-ERK signaling pathway to control cellular behavior involved in female fertility. Surprisingly while the RAS pathway has been studied extensively for decades, and much is known about the molecular architecture of the pathway, how this single pathway orchestrates a diverse array of cellular behaviors simultaneously through coordinated regulation of over two dozen effectors in one tissue remains a mystery.
We take advantage of the temporal nature of the germline in C. elegans, and through the use of phospho-substrate antibodies, and combinations of Crispr/Cas9 mediated genome engineering, we are starting to build a frame work on the molecules that define the architecture of this complex tissue. Using genetic mutants and perturbation of environmental cues, we perturb this architecture and study the behavior of the network, with the resulting impact on various meiosis I processes. Often, this results in publications on single gene effectors, but over a course of time, we expect to complete this road map and obtain a clear view of this intricate signaling society and the language they use to specify and maintain the integrity of a developing oocyte and sustenance of the next generation. Given the conserved nature of this network and the players involved, these principles may be widely applicable to various developmental systems.
In human females, oocytes complete meiosis I at birth, and enter a
long period of meiotic I and II arrest until onset of meiotic
maturation at puberty. At the end of meiosis I oocytes are packed with
maternal RNAs, which, upon fertilization of the mature oocyte, are
necessary for early embryonic development. The RNAs are degraded later
in embryogenesis to enable embryonic genome activation. Errors during
the generation and/or protection of the maternal RNAs may adversely
affect oogenesis and result in infertility. Similarly, errors in
degrading the maternal RNAs during embryogenesis can cause birth
defects. Thus, mechanisms that coordinate the protection of maternal
RNA during oogenesis with their timely degradation in the embryo are
critical for understanding the molecular basis of infertility and
birth defects; such mechanisms remain an intense area of
research.
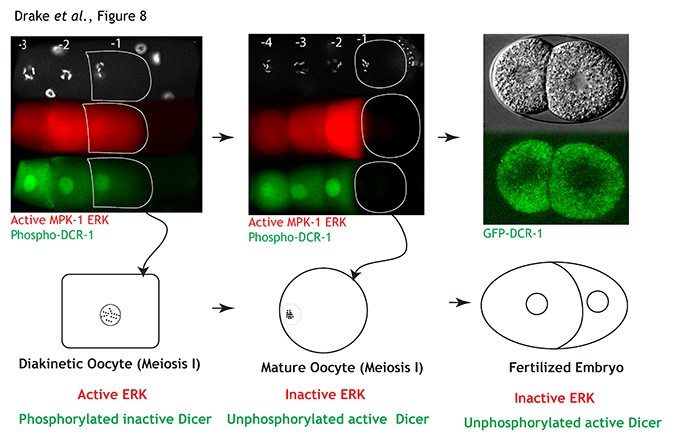
Using C. elegans meiosis I oocytes as
our model system we identified that the nutritionally regulated
RAS/ERK signaling pathway phosphorylates Dicer and Drosha, small RNA
biogenesis enzymes. Currently, we are testing the hypothesis that
signal-induced regulation of Dicer and Drosha coordinates the
protection of maternal RNAs during oogenesis with their timely
degradation in the embryo.
Current work in the lab is combining single cell oocyte transcriptomics, with high resolution imaging, small RNA profiling and developmental genetic and molecular analysis to understand these regulations.
The GTPase KRAS is an oncogene associated with tumor metastasis in many cancers. KRAS normally activates multiple signaling pathways, most notably the RAS-ERK pathway, to control cell proliferation and development, but when its function is deregulated it can drive oncogenesis and metastasis. KRAS was heralded decades ago as a major cancer drug target, but despite massive effort has proven undruggable. KRAS acts through its effector, ERK, a kinase, which phosphorylates and alters the activity of a battery of proteins. Large-scale cancer genome sequencing efforts have sought to identify gene-mutations that act with or downstream of KRAS to drive metastasis, but functional analysis on these need to be conducted to determine which ones are actionable.
By translating foundational studies in C. elegans to mammalian systems, in collaboration with Gigi Lozano, Ph.D. and her group, we discovered that phosphorylated Dicer is nuclear in mice. Homozygous phospho-mimetic Dicer mice display accelerated aging, hypermetabolism and infertility. In human tumor samples, the presence of phosphorylated, nuclear Dicer correlates with advanced, grade endometrial cancer in women. Critically, in mice heterozygous for either a p53 null allele or the constitutively active KRAS[G12D] mutation, which alone produce localized tumors, the presence of the dually phospho-mimetic form of Dicer drives cancer dissemination at high penetrance. Nuclear phosphorylated Dicer then appears to be a metastatic driver.
Studies in the lab, continued in collaboration with Lozano’s group, use a combination of genomic and proteomic analyses, with imaging-based approaches and functional analysis to understand the role of nuclear phosphorylated Dicer in mediating cancer progression.
Give Now
Research Areas
Find out about the four types of research taking place at UT MD Anderson.
