Research
The first and main aspect of research in the Vaishnavi lab focuses on understanding lung tumorigenesis at specific disease stages (shown in figure below). We are interested in dissecting how normal signaling pathways can support or modify dominant oncogenic programs at different disease stages, and with different dominant oncogenic mutations in unique mouse or human models.
Oncogene-Driven Lung Cancer Disease Progression in the Mouse Lung Model
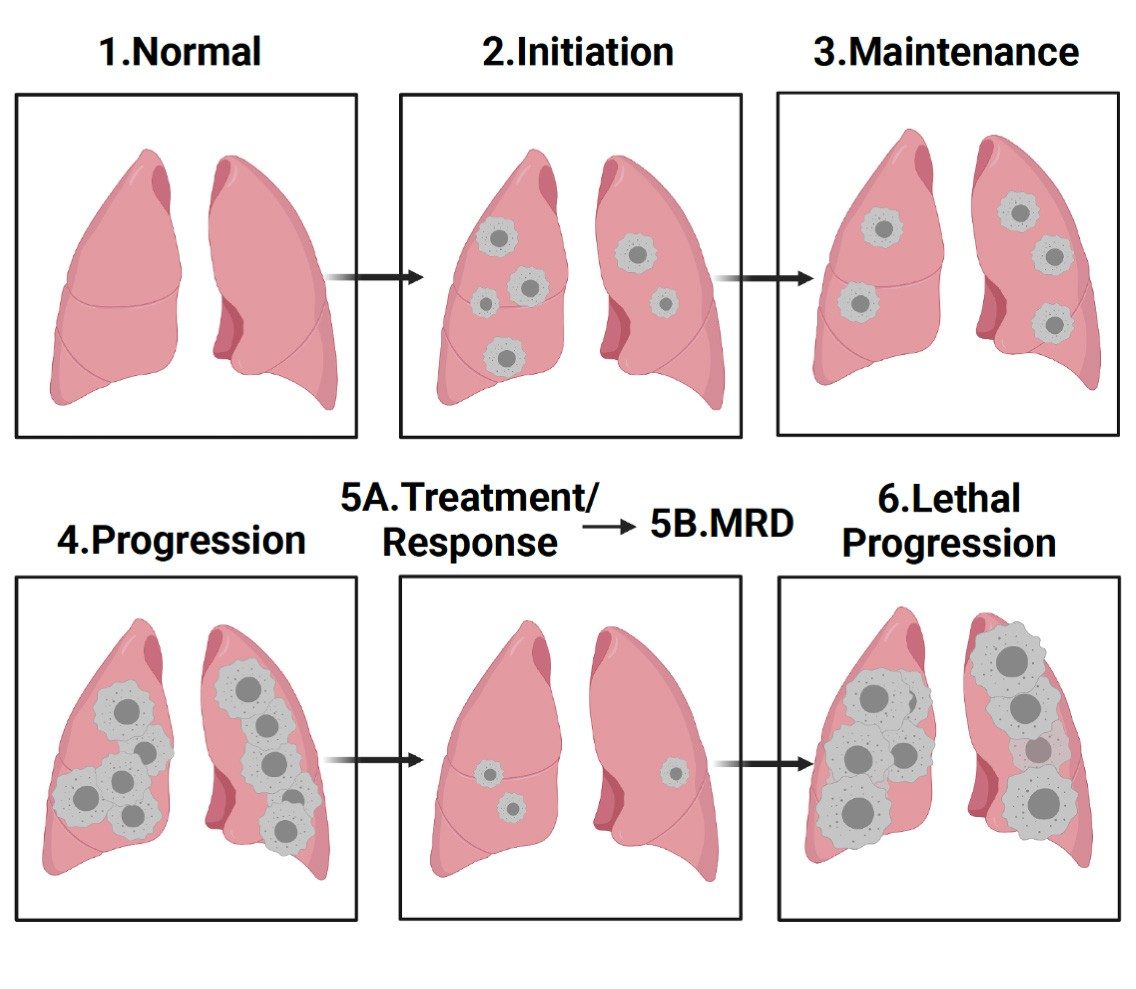
It is known that EGFR and WNT signaling are necessary for proliferation and regeneration of alveolar type 2 pneumocytes (AT2) in the lung during normal development and physiology (Desai T et Nature, 2014). Our research investigates whether after establishing an important role early in lung development, signaling from EGFR and the rest of the ERBB family that supports AT2 cell proliferation and regeneration is hijacked by the cancer. Our lab will interrogate if fundamental signaling from normal RTK pathways like the ERBB family may be contributing to support common oncogenic programs at key stages, such as initiation, maintenance, progression, and particularly: minimal residual disease. This schematic depicts tumor burden at various stages of lung tumorigenesis, and where our research will focus on elucidating unknown signaling contributions at the indicated stages. Created using BioRender.
The second aspect of our research is centered on understanding the pathways that regulate minimal residual disease (MRD) and finding ways to target them effectively. Indeed, oncogene-targeted therapies against non-small cell lung cancer (NSCLC) are powerful, improving outcomes for patients with select oncogenic mutations, including EGFR, ALK fusion kinases, or BRAF. However, our understanding of how these oncogenes work in NSCLC patients remains incomplete. Often, these benefits occur transiently, and only in certain molecular subsets. At best, oncogene-targeted monotherapy results in dramatic but incomplete tumor cell killing, and MRD. MRD are the seeds that drive therapeutic resistance, an urgent barrier to clinical success against cancer. As such, there is an urgent need to understand the pathways that contribute to MRD. We hypothesize that genes cooperating with oncogenes during initiation or progression may help establish critical survival signals during oncogene-targeted monotherapy and thus may represent important future therapeutic targets.
The third aspect of research in the Vaishnavi lab relates to studying the molecular mechanisms that underlie lung cancer in never smokers (LCINS). Chronic exposure to inhaled radon [Rn] gas is estimated by the EPA to cause 21,000 lung cancer deaths in the US annually, making it the second leading cause of lung cancer, and the leading cause of LCINS. LCINS accounts for 10-25% of all lung cancer cases, and as its own subset, accounts for one of the highest causes of cancer mortality. Radon is a noble gas naturally produced environmentally through uranium decay that releases alpha, beta, and gamma radiation. Lung injury from inhaled radon is the most common identifiable cause of LCINS. Despite this, the precise molecular, cellular and genetic changes that occur in the lung epithelium following chronic radon exposure are poorly characterized. Moreover, there are no reliable ways to identify radon-induced lung cancer in patients: a critical knowledge gap in the field. This is due, in part, to a lack of evidence directly assessing environmentally relevant levels of radon gas exposure in a controlled experimental setting. An improved understanding of the molecular mechanisms that drive radon-induced lung cancer, and the ability to identify these patients, may result in new or improved therapeutic strategies in this poorly understood disease.

Cartoon schematic depicting the dangers of air pollution and radon gas. Human environmental exposure to radon gas from the ground or fine particulate matter from air pollution can severely impact lung health, which may result in lung cancer. It has also been shown that females have a higher risk of developing what is called “lung cancer in never-smokers,” or LCINS. We will address this by exposing genetically engineered animal models to these health hazards in a novel exposure laboratory.
Give Now
Research Areas
Find out about the four types of research taking place at UT MD Anderson.
