Current Research
Ongoing funded grants
Hubert H. Chuang, M.D., Ph.D.
Role: Collaborator
Title: A Phase II Study of Ibrutinib
Goal: To evaluate the response rate of ibrutinib plus rituximab in patients with relapsed and/or refractory MCL.
Homer A. Macapinlac, M.D.
Role: Co-Investigator
Title: Evaluation of Interim FLT-PET/CT for Early Prediction of Outcome of Patients with Diffuse Large B-cell Lymphoma (DLBCL) Treated with R-CHOP
Goal: To demonstrate of superiority of FLT-PET/CT over FDG-PET/CT, which is expected to lead to greater confidence in using interim PET in patient management.
Role: Co-Investigator
Title: Tumor mutation status will predict metabolic response to metformin in NSCLC.
Goal: To predict metabolic response to metformin in NSCLC.
Role: Co-Investigator
Title: Southwest Early Clinical Trials Consortium
Goal: To strategically and geographically collaborate on and execute phase I clinical trials in the NCI Experimental Therapeutics-Clinical Trials Network in broad patient populations, including several underserved regions.
Role: Collaborator
Title: Program Project 2: Molecular Imaging of EGFR Expression and Activity in Targeted Therapy of Lung Cancer
Goal: To develop molecular imaging techniques to identify and quantify EGFR expression and activity in lung cancer.
Role: Primary Investigator
Title: Multi-Modal Reconstruction and Quantitative SPECT Imaging
Goal: To evaluate the feasibility of applying the novel multimodal reconstruction technique for SPECT/CT imaging in oncology patients.
Related Departments & Divisions
Nuclear Medicine Highlights
Whole Body PET MIP Images
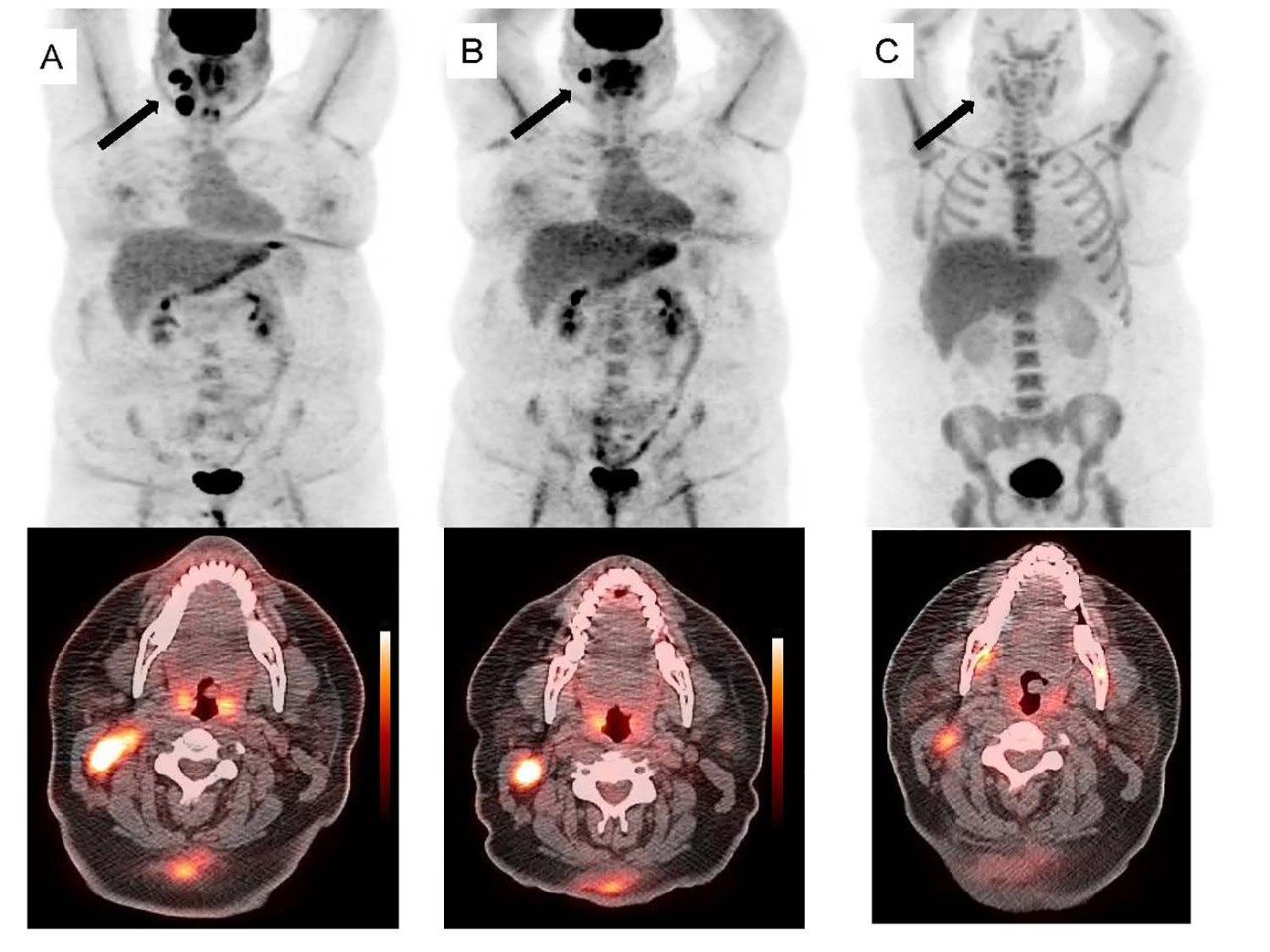
Figure 1, top row: whole body PET maximal intensity projection (MIP) images; lower rows: transaxial PET/CT fusion images of the neck)
Baseline: FDG PET and PET/CT shows intense FDG uptake (SUVmax=16.8) in an enlarged right cervical lymph node (arrow).
B: Interim FDG PET and PET/CT shows volume reduction of the lymph node (arrow) but SUVmax increased (SUVmax=21.5).
C: Interim FLT PET and PET/CT shows visually positive uptake in this lymph node (arrow). This was confirmed as residual lymphoma.
Baseline FDG PET MIP images

Figure 2
Baseline FDG PET MIP image showing intense FDG uptake in a bulky mass within the left pelvis (arrow).
B: Interim FDG PET MIP image demonstrating significantly decreased activity but remained positive (arrow).
C: Interim FLT PET, shows photopenic tracer uptake in this lesion, reported as negative for disease.
D: End-of-treatment FDG PET MIP image shows significant reduction in FDG activity and classified as complete response based on both imaging and clinical outcome, confirming FLT PET/CT result.
FDG PET/CT Images

Figure 3
Baseline FDG PET/CT (upper panel: transaxial PET/CT fusion image of the pelvis, lower panel: transaxial PET image) showing intense FDG activity at the left ilium (arrow).
B: Interim FDG PET/CT (upper panel: transaxial PET/CT fusion image of the pelvis, lower panel: transaxial PET image) demonstrates complete response at the lesion site.
C: Interim FLT PET/CT (upper panel: transaxial PET/CT fusion image of the pelvis, lower panel: transaxial PET image) shows absence of tracer uptake within the osseous lesion. Clinical outcome confirmed this case as complete response.
Give Now
Research Areas
Find out about the four types of research taking place at UT MD Anderson.
