3D Modeling of the Tumor Ecosystem
Research
Research within the Center for Cellular Language Intelligence (CCLI) is organized around clinically grounded, end-to-end discovery and translation. Starting with high-impact clinical questions, CCLI integrates spatial profiling, computational modeling, artificial intelligence and functional validation to unravel the spatial principles and signaling hubs that govern tumor ecosystems, influencing tumor behavior, immune response and clinical outcomes.
By integrating these capabilities within a single framework, the center moves beyond descriptive atlases toward causal understanding and clinically actionable insight, uncovering the signals and control hubs that drive tumor behavior and converting those insights into actionable biomarkers, therapeutic targets and new strategies to improve patient care.
This video (with no sound) shows a model of the tumor ecosystem.
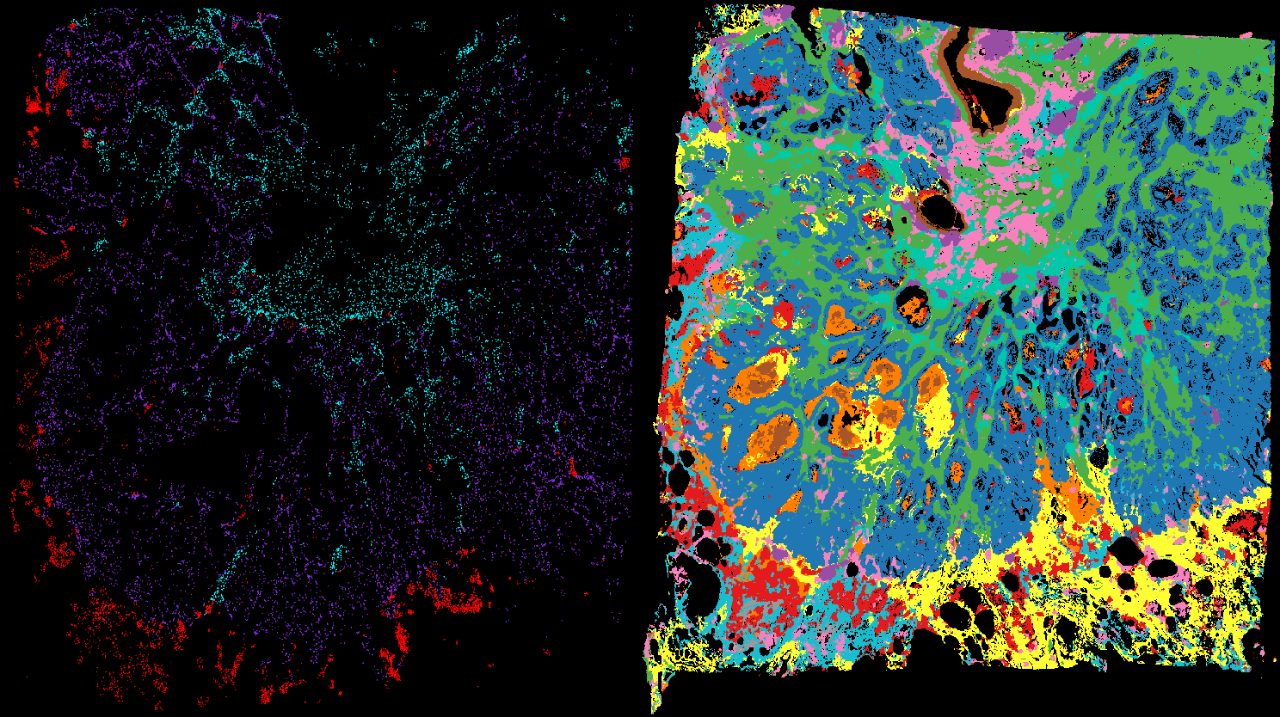
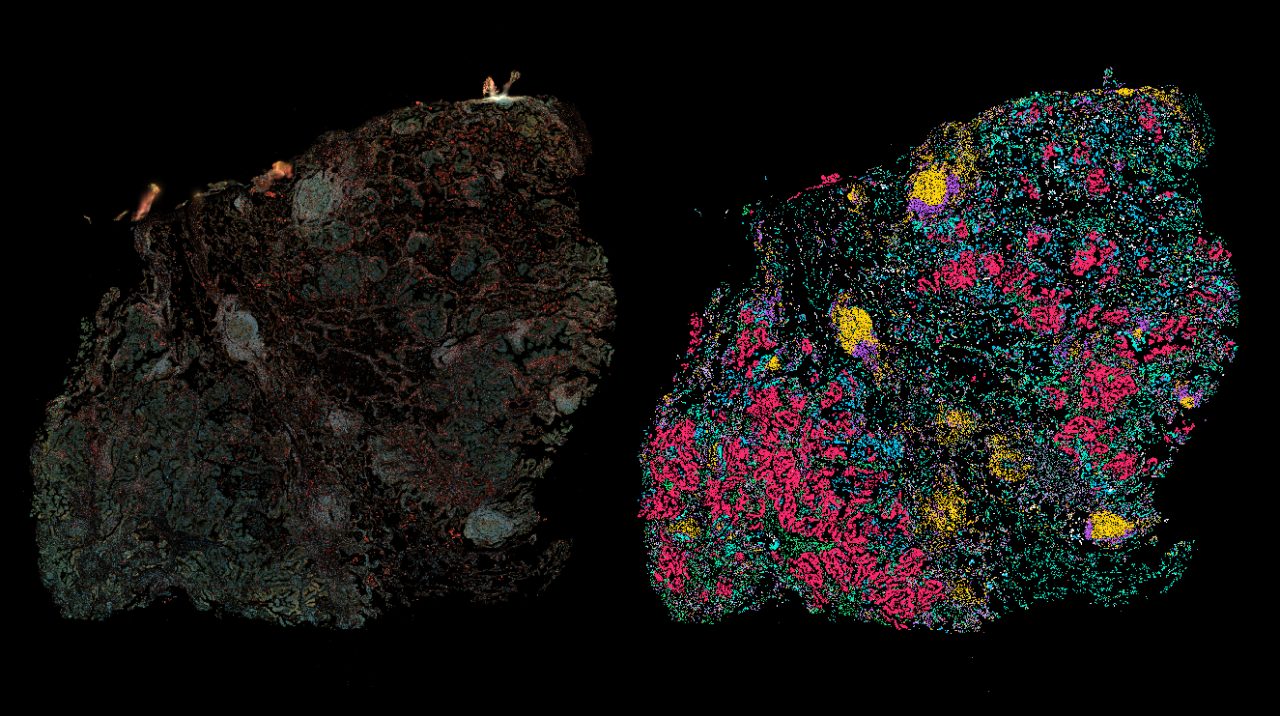
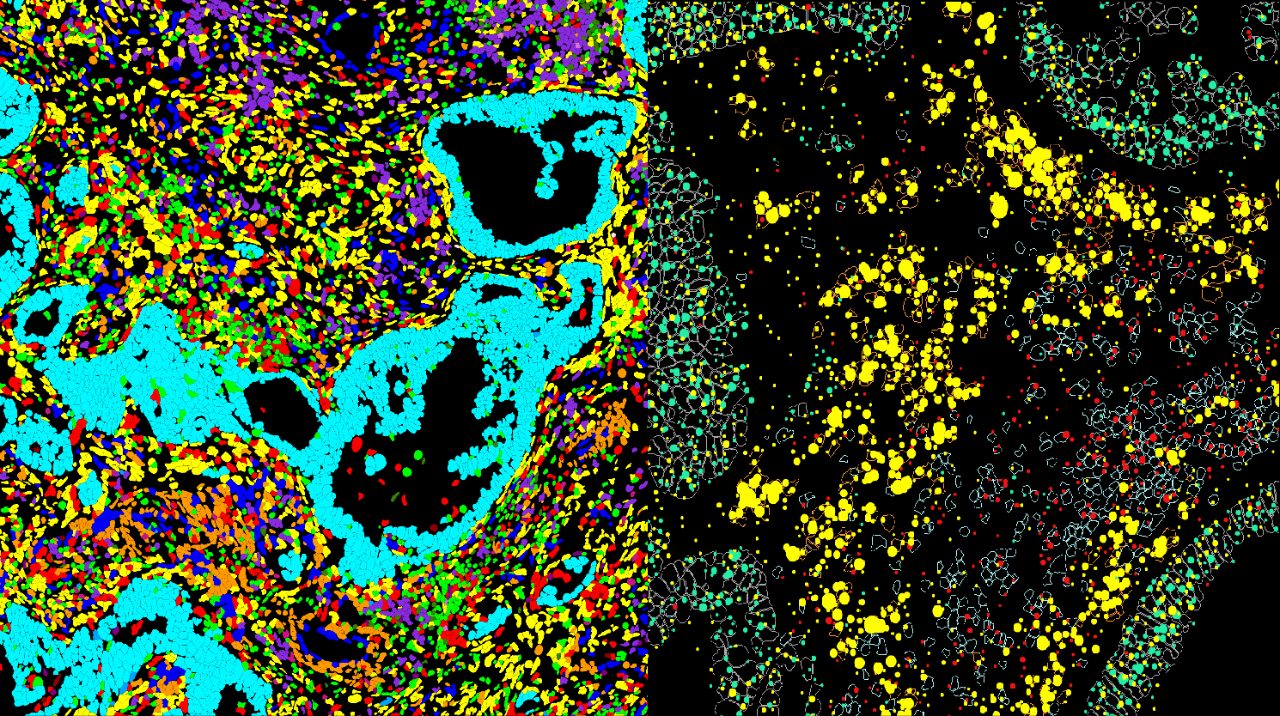


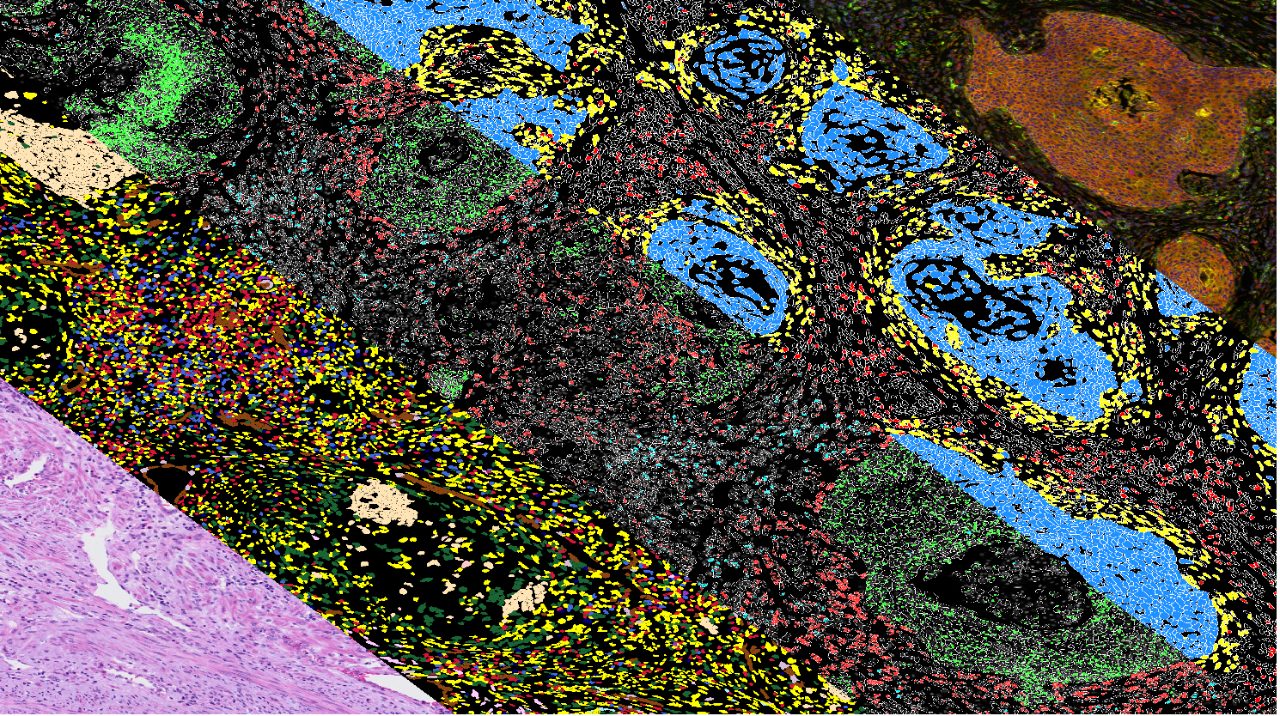
Research Teams
Overview
The center is organized into coordinated functional teams that span its end-to-end discovery and translation workflow — from spatial ecosystem profiling and computational modeling to biomarker and target discovery, functional validation and clinical translation. Together, these teams enable the center to decode how cells organize, communicate, function and adapt within the tumor ecosystems across space and time and to translate those insights into actionable advances that improve cancer prevention, diagnosis and treatment.
Spatial Profiling
The Spatial Ecosystem Profiling Team specializes in research specimen handling, pathology review, tissue annotation, spatial assay optimization and high-resolution spatial multimodal profiling. The team will also rapidly evaluate, adopt and optimize emerging spatial technologies as they become available, ensuring that the center remains at the forefront of innovation in spatial biology.
This team generates high-quality histology, spatial multi-omics and 3D/4D data from patient specimens and experimental models, creating the foundational datasets that power the center’s discovery engine. By standardizing workflows across platforms and ensuring rigorous quality control, the team helps generate high-quality, clinically informative data that can be compared and integrated across cohorts, modalities and studies.
Computational Modeling & Analytics
The Computational Modeling & Analytics Team serves as the center’s analytical engine, applying advanced computational approaches to transform complex spatial and multimodal datasets into biological and clinical insights. By integrating spatial multi-omics (DNA, RNA, protein, metabolomics, etc.), molecular imaging, histology and clinical data, the team characterizes tissue architecture and intercellular interactions; maps cell states, functional niches, spatial communities and signaling hubs; and reveals the organizational principles that shape tumor behavior, immune response and clinical outcomes.
Leveraging state-of-the-art computational methods, 3D spatial reconstruction, emerging foundation models, artificial intelligence (AI)/machine learning (ML) tools and AI agents, the team uncovers candidate drivers, biomarkers, signaling hubs and predictive features associated with cancer initiation, progression, therapeutic response and resistance.
The team also curates and integrates data from in-house cohorts and published studies, harnessing the power of big data, knowledge bases and AI co-scientists to find signals in hyper-dense tumor communication networks, generate and test hypotheses in curated datasets and help identify high-probability drivers. It develops interpretable and mechanism-informed models to support biomarker development, patient stratification and clinical translation.
AI Biomarker & Target Discovery
The AI-Enabled Biomarker & Target Discovery Team focuses on translating complex multimodal data into clinically actionable biomarkers and therapeutic targets. Building on insights generated across spatial profiling and computational modeling, the team integrates data across modalities (including genomics, transcriptomics, proteomics, metabolomic, imaging and clinical data) and across scales (from molecular and cellular states to tissue architecture and ecosystem-level features) to identify robust, reproducible signals associated with cancer risk, progression and therapeutic response. The team prioritizes cancer-specific and conserved biomarkers and targets across cancer types that are aligned with critical clinical needs, including early detection, patient stratification, drug target discovery and prediction of treatment response and resistance.
Leveraging advanced AI/ML frameworks, foundation models and virtual tumor ecosystem representations, the team also models tumor ecosystems in silico and linking these models to real-world clinical and molecular data to enable hypothesis-driven prediction of drug response and support the identification of high-confidence therapeutic opportunities.
Through close integration with functional genomics and clinical translation efforts, this team helps bridge discovery and application, ensuring that identified biomarkers and targets are both biologically grounded and positioned for clinical validation and deployment.
Functional Genomics & Validation
The Functional Genomics & Experimental Validation Team bridges computational discovery with mechanistic interrogation to move from correlation to causality. Building on prioritized biomarkers, targets and ecosystem features identified by upstream teams, this group designs and executes high-throughput and targeted functional studies to test biological hypotheses and validate candidate drivers.
Leveraging CRISPR-based perturbation strategies, spatially informed functional assays, organoid systems and other human-relevant experimental models, the team systematically evaluates gene function, regulatory programs and cell-cell interaction mechanisms within the tumor ecosystem. These approaches enable precise dissection of the molecular and cellular processes that underlie tumor initiation, progression, immune modulation and therapeutic response or resistance.
Through iterative integration with computational modeling and AI-driven discovery, the team refines candidate targets and prioritizes those with the highest biological relevance and translational potential. This work ensures that discoveries emerging from the center are not only statistically robust, but also mechanistically grounded and positioned for downstream clinical translation.
Clinical Translation
The Clinical Translation Team ensures that discovery is tightly connected to patient care. The team helps anchor center projects in clinically meaningful questions, supports strategic cohort design and aligns biospecimen collection, clinical metadata and study endpoints with translational goals. By integrating clinical insight from the outset, the team helps ensure that the center’s research remains closely aligned with pressing clinical needs.
Working across clinical and research domains, the team facilitates the flow of biospecimens, data and clinical context needed to support high-impact translational studies. It also helps position promising biomarkers, targets and predictive models for clinical validation, trial integration and eventual deployment in precision oncology.
Through these efforts, the Clinical Translation Team serves as a critical bridge between scientific discovery and patient benefit.
Give Now
Research Areas
Find out about the four types of research taking place at UT MD Anderson.
