Research
Featured Research
Versatile Platform for Capture & Analysis of Rare Cells

We have developed a versatile assay for highly efficient capture of circulating cancer cells. The methodology is based on hybrid magnetic/plasmonic nanocarriers and a novel microfluidic chamber (top left cartoon.) In this assay cancer cells are specifically targeted by antibody-conjugated magnetic nanocarriers and are separated from normal blood cells by a magnetic force in a microfluidic chamber.
Subsequently, immunofluorescence staining is used to differentiate CTCs from normal blood cells (bottom left figure.) Alternatively, the captured CTCs can be detected using photoacoustic (PA) imaging with near-infrared (NIR) excitation and, then, collected for a downstream analysis.
We demonstrated in cell models of colon, breast and skin cancers that this platform can be easily adapted to a variety of biomarkers, targeting both surface receptor molecules and intracellular biomarkers of epithelial-derived cancer cells. Experiments in whole blood showed capture efficiency greater than 90% when two cancer biomarkers are used for cell capture (right graph.)
Thus, the combination of immunotargeted magnetic nanocarriers with microfluidics provides a promising platform that can improve the effectiveness of current CTC assays by overcoming the problem of heterogeneity of tumor cells in the circulation.
For more details see: Wu CH, Huang YY, Chen P, Hoshino K, Liu H, Frenkel EP, Zhang JXJ, Sokolov KV. Versatile Immunomagnetic Nanocarrier Platform for Capturing Cancer Cells, ACS Nano v. 7(10): 8816-8823, 2013.
Imaging and Tracking of Single Biomolecules

Polarization microscopy with stellated gold nanoparticles for in-situ monitoring of biomolecules. The movie on the left shows interactions between individual ca. 40nm EGFR-targeted stellated gold nanoparticles and a single SiHa cervical cancer cell. The images are obtained in a dark-field reflectance mode through cross-polarizers that suppresses scattering signal from the cell.
In the movie on the right the dark-field images are overlaid with a phase contrast image taken from the same field of view to show cell location. Note the presence of unbound particles moving quickly through the field of view. Bound particles show slower, directional movement consistent with receptor binding.
The efficient light depolarization by stellated gold nanoparticles allows robust detection of biomolecules in cross-polarized imaging where the intrinsic light scattering from cells is significantly reduced. The imaging can be carried out with single molecule sensitivity for essentially unlimited time with no signal degradation.
For more details see: Aaron J, de la Rosa E, Travis K, Harrison N, Burt J, José-Yacamán M, Sokolov K, Polarization Microscopy with Stellated Gold Nanoparticles for Robust, In-Situ Monitoring of Biomolecules. Optics Express v. 16(3): 2153-2167, 2008.
“Stealth” Nanoparticles – The Concept of a Hydrophobic Shield

Polyethylene glycol (PEG) surface coatings are widely used to render stealth properties to nanoparticles in biological applications. However, physiological concentrations of cysteine and cystine can displace methoxy-PEG-thiol molecules from the gold nanoparticle (GNP) surface that leads to protein adsorption and cell uptake in macrophages within 24 hours (schematic illustration top row).
We addressed this problem by incorporating an alkyl linker between the PEG and the thiol moieties that provides a hydrophobic shield layer between the gold surface and the hydrophilic outer PEG layer. The mPEG-alkyl-thiol coating greatly reduces protein adsorption on GNP-alkyl-PEG particles and their macrophage uptake (schematic illustration bottom row). This has important implications for the design of gold nanoparticles for biological systems.
The transmitted brightfield images show cells incubated for 24 hours with either GNP-PEG (top row) or GNP-alkyl-PEG (bottom row). The nanoparticles were pre-incubated in complete media for 1 day, 3 days and 5 days. The presence of nanoparticles is readily apparent in cells treated with GNP-PEG as a dark contrast due to light absorption by the particles, while there is no detectable particles visible in the GNP-alkyl-PEG samples.
Scale bar is ca. 20 um.
For more details see: Larson TA, Joshi PP, Sokolov K. Preventing Protein Adsorption and Macrophage Uptake of Gold Nanoparticles via a Hydrophobic Shield, ACS Nano v. 6(10): 9182-9190, 2012.
Dynamic Imaging of Molecular Assemblies
We have pioneered a new method for imaging of dynamic behavior of molecular assemblies in-situ. The method is based on nanoparticle plasmon resonance coupling (NPRC) in combination with hyperspectral optical imaging and statistical image analysis.
A primary strength of NPRC as a bio-sensing tool is associated with the complex optical behavior of plasmonic nanoparticle assemblies.
Strong changes in optical properties associated with nanometer-scale changes in 3D organization of biomolecules facilitate development of statistical associations with the biological processes under observation.
The method is finding applications in studies of signaling dynamics in live cells, in drug screening assays, molecular detection and in molecular therapy monitoring.
For more details see: Aaron J, Travis K, Harrison N, Sokolov K. Dynamic Imaging of Molecular Assemblies in Live Cells Based on Nanoparticle Plasmon Resonance Coupling. Nano Letters v. 9(10): 3612-3618, 2009.
Nanotheranostics

Our lab has been developing multi-modal magneto-plasmonic nanoparticles that can be used with multiple imaging modalities including optical (left: dark-field image of EGFR-expressing cancer cells), MRI (T2-weighted images of various concentrations of labeled EGFR(+) cells; detection limit ca. 30-40 labeled cells per 0.0225 mm3 voxel with 4.7T system), photoacoustic (not shown) molecular imaging and photothermal cancer therapy.
For more details see:
a. Larson TA, Bankson J, Aaron J, Sokolov KV. Hybrid plasmonic magnetic nanoparticles as molecular specific agents for MRI/Optical imaging and photothermal therapy of cancer cells. Nanotechnology 18, 325101:8 pp, 2007
b. Larson T, Travis K, Joshi P, Sokolov K, Nanoparticles for Targeted Therapeutics and Diagnostics. In: Handbook of Biomedical Optics, Eds. Boas, Pitris and Ramanujam, Taylor & Francis Books, Inc., pp. 697-721, 2011.
Nanotherapeutics

We have shown that multivalent presentation of therapeutic antibodies (Abs) can influence their biological function.
We demonstrated that conjugates of multi-modal magneto-plasmonic nanoparticles with anti-EGFR antibody (cetuximab, C225) synergistically induce autophagy and apoptosis in non-small cell lung cancer cells and significantly increase therapeutic efficacy (ca. 3-fold) as compared to the same amount of free antibodies; no autophagy was induced by free antibodies.
We also showed potent in vivo antitumor activity of C225-AuNP conjugates in a lung xenograft cancer model which had no growth inhibition response to treatment with free C225 antibodies.
For more details see:
Yokoyama T, Tam J, Kuroda S, Scott AW,
Aaron J, Larson T, Shanker M, Correa AM, Kondo S, Roth JA, Sokolov K*,
Ramesh R.* EGFR-targeted
hybrid plasmonic magnetic nanoparticles synergistically induce
autophagy and apoptosis in non-small cell lung cancer cells,
PLoS ONE v.6(11): e25507, 13 pages, 2011 (*corresponding authors)
Biodegradable Plasmonic Nanoparticles for Cancer Imaging and Therapy

Figure. The concept of a plasmonic nanocluster that biodegrades in a slightly acidic environment of cellular endosomal compartments.
A fundamentally new approach to synthesis of plasmonic nanomaterials for biomedical applications and clinical translation that is based on assembly of primary nanoparticles with predetermined properties to nanoclusters of well-defined size using a biodegradable polymer.
The nanomaterial exhibits a new range of properties including biodegradation, strong near-infrared NIR absorbance at a very small size, greater photostability, and stronger photoacoustic signal as compared to previously known plasmonic nanoparticles.
Key Collaborators: K. Johnston and S. Emelianov, the University of Texas at Austin
Funding: NIH/NCI R01
Capture, Enumeration and Characterization of Circulating Tumor Cells

Figure: Conceptual cartoon of the versatile immunomagnetic nanocarrier platform in microfluidics for capturing circulating tumor cells in whole blood.
Detection of disseminated tumor cells (DTC) or tumor markers (TMs) in human fluids such as blood, urine, saliva, and sputum can provide an opportunity to develop an accessible screening tool for cancer detection and can help in determining prognosis and monitoring the effectiveness of therapy.
However, the technical challenge to identifying CTCs in whole blood is their extremely small abundance - just about one CTC per 106 - 109 blood cells.
Our goal is to develop a highly sensitive, low cost, one-step assay that would allow detection and characterization of cancer cells in a variety of unpurified human specimens, such as blood.
To achieve this objective we explore approaches that are based on recent technological advances in immunotargeted hybrid magneto-plasmonic nanocarriers, photoacoustic (PA) imaging as well as arrays of metamaterial-based plasmonic nanosensors.
Key Collaborators: J. Reuben and R. Alvarez, MD Anderson Cancer Center; G. Shvets, S. Emelianov and J. Zhang, the University of Texas at Austin; E. Frenkel, UT Southwestern Medical Center.
Funding: NIH/NCI R01, R21
Optical Systems for In Vivo Molecular Imaging of Cancer

The goal of this NCI supported Bioengineering Research Partnership which combines five performance sites including MD Anderson is to optimize and to translate contrast agents and imaging systems for early diagnosis of pre-cancer in the upper aero-digestive tract, culminating with pilot in vivo studies in human patients to assess sensitivity and specificity.
Figure: Molecular imaging of EGFR-expressing epithelial pre-cancers in the oral cavity using fluorescently labeled anti-EGFR antibodies (left column) and anti-EGFR plasmonic nanoparticles (right column). The top row shows normal tissue and the bottom row – pre-cancerous tissue.
The plasmonic nanoparticles provide ca. 5 fold additional increase in contrast between normal and abnormal tissue due to the effect of plasmon resonance coupling that is mediated by specific interactions with EGFR receptors.
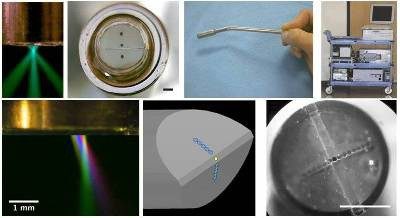
Figure: A spectroscopic fiber optic probe for sensitive and specific detection of precancerous lesions in the oral cavity using oblique polarized reflectance spectroscopy (OPRS).
Key Collaborators: R. Richards-Kortum, Rice University; A. Gillenwater, L. Coghlan and J. Lee, MD Anderson Cancer Center; C. McAulay, British Columbia Cancer Agency.
Funding: NIH/NCI R01
Give Now
Research Areas
Find out about the four types of research taking place at UT MD Anderson.
