UT MD Anderson names Albert Koong, M.D., Ph.D., chief scientific officer
The University of Texas MD Anderson Cancer Center today announced that Albert Koong, M.D., Ph.D., an internationally recognized physician-scientist, has been appointed chief scientific officer (CSO), effective June 1. Koong has served as CSO ad interim since Jan. 1.
The CSO role oversees UT MD Anderson’s laboratory and clinical research enterprise, champions innovation, develops strong partnerships, and provides focused leadership...


CPRIT awards UT MD Anderson over $19 million for cancer research and faculty recruitment
The University of Texas MD Anderson Cancer Center today was awarded over $19 million from the Cancer Prevention and Research Institute of...
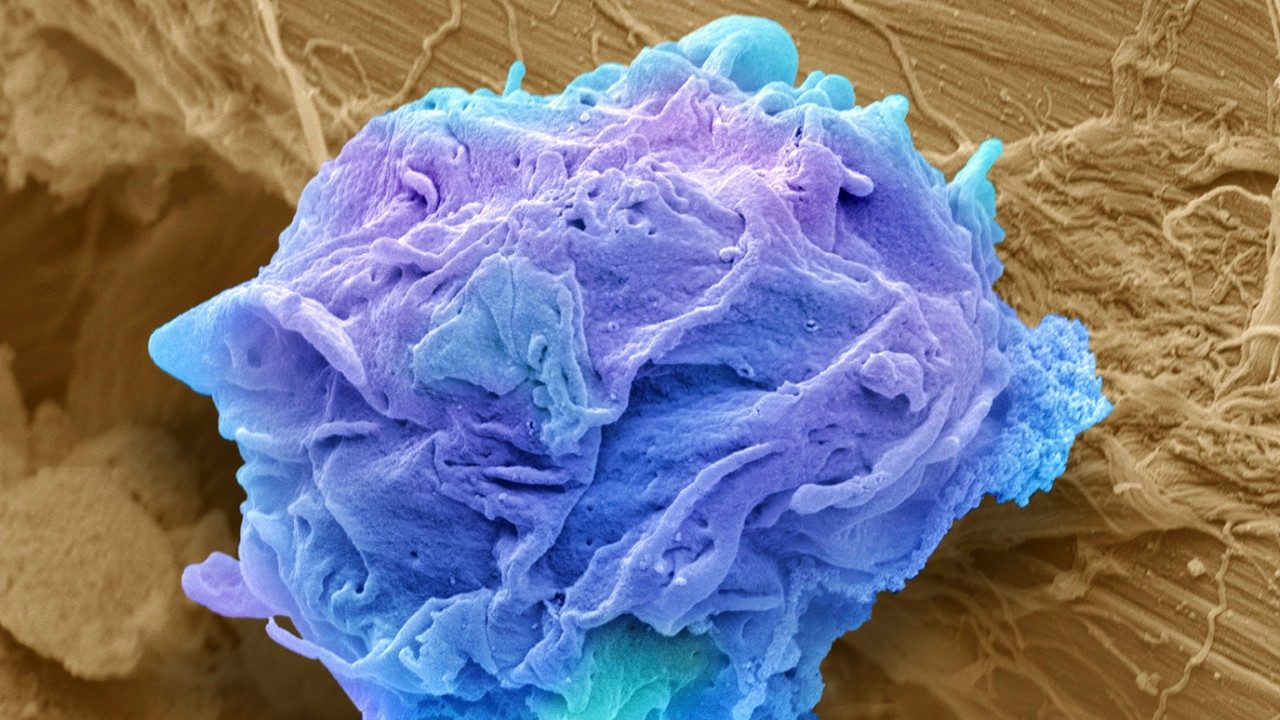
Novel CAR T cell therapy developed by UT MD Anderson researchers moves into clinical studies
UT MD Anderson appoints Kim Slusser as chief nurse executive
The University of Texas MD Anderson Cancer Center today announced the appointment of Kim Slusser, Ph.D., R.N., as the institution’s inaugural chief nurse executive (CNE), effective May 1.
As CNE, Slusser will serve as the enterprise-wide leader of more than 5,400 nurses, fostering a shared vision for high-quality nursing care. She will oversee strategic initiatives spanning clinical practice, operations, quality, safety, emergency...


UT MD Anderson establishes Center for Cellular Language Intelligence with $10 million gift from Peggy and Carl Sewell
The University of Texas MD Anderson Cancer Center today announced a $10 million gift from Peggy and Carl Sewell to...

AACR: Jennifer Wargo, M.D., elected Fellow of the AACR Academy
Jennifer Wargo, M.D., professor of Surgical Oncology and Genomic Medicine at The University of Texas MD Anderson...

AACR: Immunotherapy pioneer James P. Allison, Ph.D., honored with Award for Lifetime Achievement in Cancer Research
James P. Allison, Ph.D., Regental professor and chair of Immunology, vice president of immunobiology and founding director of the James P....

Two UT MD Anderson researchers elected AAAS Fellows
In recognition of their significant achievements to advance cancer care and research, Theresa Guise, M.D., and Stephanie S. Watowich, Ph.D...

UT MD Anderson opens new Colorectal Center to provide specialized care in healing environment
The University of Texas MD Anderson Cancer Center announced the opening of its new Colorectal Center, located on...

Peter WT Pisters, M.D., honored with Charles M. Balch, M.D., Distinguished Service Award from Society of Surgical Oncology
Top award from Society of Surgical Oncology honors physicians, scientists and physician-scientists who have made outstanding...
