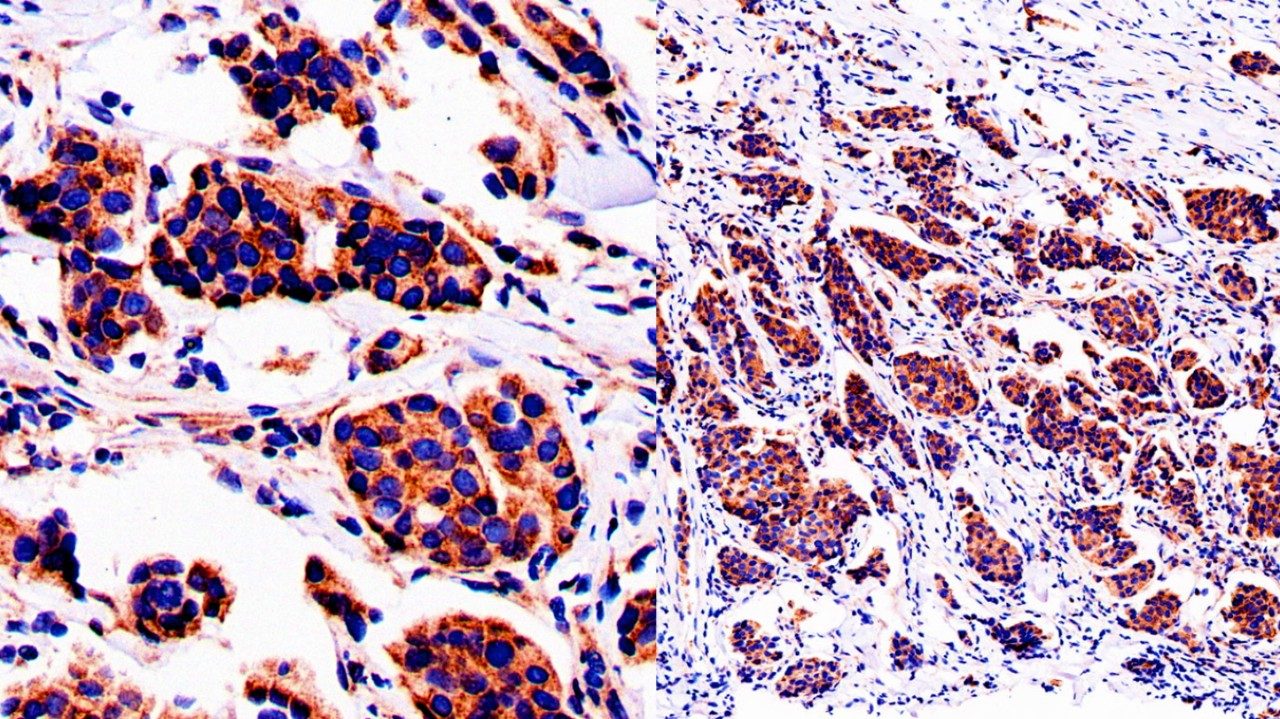
Swollen collar bone (supraclavicular) lymph nodes: 4 things to know
You may already be aware that swollen lymph nodes can be a sign of cancer. But did you also know that where they arise on the body can sometimes hint at which type of cancer is causing them?
With oral cancers, for instance, swollen lymph nodes tend to appear first in the neck or under the jaw. Breast cancer, on the other hand, tends to affect the lymph nodes in the armpit, or axilla, first.
But what about supraclavicular lymph nodes — the ones located just above your collarbones (the clavicles)? What does a swollen lymph node in the collarbone area mean, and when should you see a doctor about it?
We asked lymphoma specialist Luis Fayad, M.D. for insight. Here’s what he shared.
Swollen lymph nodes can be a sign of many different types of cancer
The first thing to know about swollen supraclavicular lymph nodes? They can be a sign of many different cancers, including late-stage:
- Breast cancer
- Esophageal cancer
- Genitourinary cancers: including bladder, kidney and prostate
- Hodgkin lymphoma
- Lung cancer
- Melanoma
- Pelvic cancers: including cervical, endometrial and ovarian
- Stomach cancer
“Lymphoma is probably the most common diagnosis we see in patients with that symptom here at UT MD Anderson,” notes Fayad. “But I have seen cases in which the disease turned out to be ovarian, breast or lung cancer.”
Persistent fevers, unexplained weight loss and night sweats are other symptoms typically seen when the underlying disease is lymphoma.
Cancer is not the only cause of swollen supraclavicular lymph nodes
The second thing to know is that cancer isn’t the only possible reason you might have swollen supraclavicular lymph nodes.
“You can have an enlarged lymph node in that area without it being malignant,” notes Fayad. “It could be benign.”
Why? Because it could also be a sign of an inflammatory disorder or an autoimmune disease, such as:
- Lupus, also known as systemic lupus erythematosus
- Sjögren’s disease
- Rheumatoid arthritis
- Sarcoidosis
- Many others
It could even be caused by a condition like mastitis, when the milk ducts of the breast become inflamed and infected during breastfeeding, or something like a cyst or a lipoma, a benign fatty tumor.
“The collar bone is not a common location for an infection to affect the lymph nodes, though, so it’s more suspicious than it would be if it was in your neck and you also had a sore throat,” says Fayad.
When to see a doctor about swollen supraclavicular lymph nodes
So, when should you see a doctor about swollen lymph nodes on your collar bone? Here are three questions to ask yourself:
- Is the lymph node painful? “Infections are usually painful, while malignancies are not,” Fayad explains.
- Is the lymph node moveable or stationary? “Infected lymph nodes tend to be mobile, while cancerous lymph nodes tend to be fixed to the underlying tissue,” he says.
- How long has it been there? “If it doesn’t go away in a month or so, ask your doctor for a biopsy,” suggests Fayad. “And, if it persists, despite treatment with antibiotics or steroids, seek out a specialist.”
Many advanced cancers can be treated, if not cured
Even if a swollen lymph node on your collar bone turns out to be cancerous, it’s important to stay positive.
“Some of these cancers can be cured, including lymphomas, even if they’re advanced,” Fayad notes.
Request an appointment at UT MD Anderson online or call 1-877-632-6789.
Finding the right therapist for you: 3 tips from our experts
Swollen collar bone (supraclavicular) lymph nodes: 4 things to know
Elastography 101: How this test helps diagnose liver disease
Metastatic melanoma caregiver, employee encourages blood donation
After a brachytherapy clinical trial, stage 4 melanoma is now cancer-free
Phase 1 clinical trials, explained: 11 things to know
|
$entity1.articleCategory
|
|---|
|
$entity2.articleCategory
|
|
$entity3.articleCategory
|
|
$entity4.articleCategory
|
|
$entity5.articleCategory
|
|
$entity6.articleCategory
|
Find stories by topic
Find out everything you need to know to navigate a cancer diagnosis and treatment from UT MD Anderson’s experts.
Read inspiring stories from patients and caregivers – and get their advice to help you or a loved one through cancer.
Get UT MD Anderson experts’ advice to help you stay healthy and reduce your risk of diseases like cancer.
Learn how UT MD Anderson researchers are advancing our understanding and treatment of cancer – and get to know the scientists behind this research.
Read insights on the latest news and trending topics from UT MD Anderson experts, and see what drives us to end cancer.
Find out what inspires our donors to give to UT MD Anderson, and learn how their generous support advances our mission to end cancer.