After a stem cell transplant, professor returns to the classroom
Chemistry professor Bindu Chakravarty rarely missed a day teaching at Houston Community College. So when she called in sick one morning, it was with good reason.
“I had a 101-degree fever and was so exhausted, I could barely get out of bed,” she remembers. “Something was very wrong.”
For several weeks, Chakravarty, 59, had been fatigued and running a low-grade fever. But she took aspirin and kept on going.
“I thought whatever illness I was fighting would run its course,” she says. “I didn’t want to let my students down.”
When aspirin ceased to work and her fever spiked, Chakravarty’s worried husband insisted she visit a doctor. Instead of going to class, she headed to her family physician’s office.
A blood test revealed her platelets — cells in the blood that help it clot — were low. The doctor explained that this can cause excessive bruising and bleeding.
“Now I understood why my gums bled when I brushed my teeth,” Chakravarty recalls, “and why I was covered in bruises.”
Because a low platelet count can be a red flag for leukemia, Chakravarty’s doctor referred her to an oncologist at MD Anderson, where tests revealed she had a cancer of the blood called acute myeloid leukemia, or AML.
“I was admitted to the hospital the next day,” Chakravarty says, “and my cancer journey began.”
Out of control
AML starts in the soft, spongy bone marrow, where blood is made. Immature, primitive stem cells inside the marrow evolve over time into three types of mature, healthy blood cells — white to fight infections, red to carry oxygen throughout the body, and platelets to help the blood clot. But in AML, the marrow produces too many immature white blood cells, called leukemic blasts.
Because they fail to develop properly, the leukemic blasts can’t fight infections. And because so many are being produced, they crowd the bone marrow and prevent it from making the normal platelets and red and white blood cells the body needs.
“Though AML starts in the bone marrow, it can quickly move into the blood and sometimes spread to other parts of the body, including the lymph nodes, liver, brain and spinal fluid,” says Elizabeth Shpall, M.D., deputy chair of Stem Cell Transplantation and Cellular Therapy at MD Anderson. “It’s important to start treatment soon after diagnosis.”
The treatment plan
Early tests at MD Anderson revealed Chakravarty had a form of AML that’s linked to a genetic mutation and is particularly aggressive.
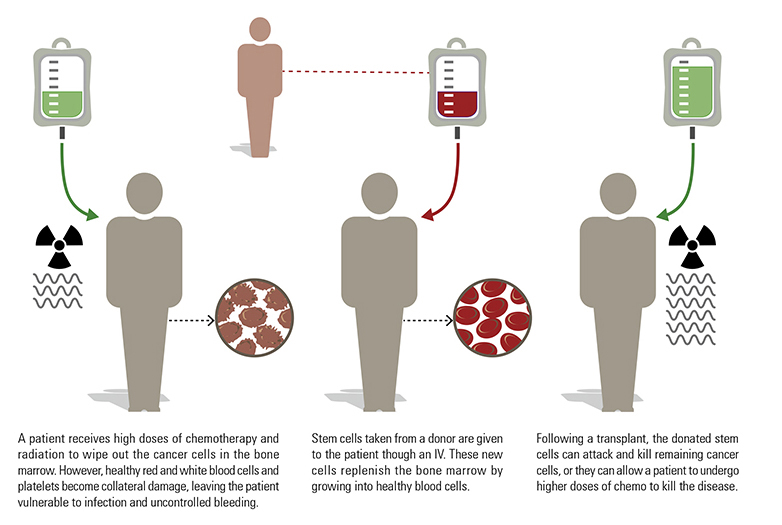
Treatment began immediately. First, doctors in the Leukemia Department prescribed several rounds of intense chemotherapy to wipe out the cancer cells in her bone marrow.
The treatment can be effective, but it has an unwanted side effect: Healthy red and white blood cells and platelets become “collateral damage” and are killed along with the cancer cells by the chemo. This makes the body vulnerable to infection and uncontrolled bleeding.
For her own protection, Chakravarty spent 27 days in isolation until tests showed no remaining signs of cancer. Next came the second phase of treatment — a stem cell transplant to replenish her bone marrow with healthy stem cells from a matching donor.
To test whether a donor is a suitable match for a recipient, doctors examine genes in the human leukocyte antigen, or HLA System — the part of the immune system that recognizes self and not self. In a full match, eight or more of the HLA genes need to match between donor and recipient. When stem cells are derived from another source — donated umbilical cord blood — a match in four of six HLA markers is considered acceptable for most patients.
None of Chakravarty’s family members qualified as a match, nor did any of the 28 million people who signed up as donors with various registries. So Shpall recommended using blood collected from an umbilical cord immediately following the delivery of a baby.
After the umbilical cord is cut, a needle is inserted into the vein of the cord, and the blood in the cord and placenta is collected and frozen until it’s needed for a transplant.
This infant blood is rich in new stem cells that haven’t yet been educated against foreign invaders, reducing the chances they’ll attack a recipient’s tissues.
“The nice thing about cord blood is it contains a large number of stem cells that are naïve,” Shpall explains. “So they’re less likely to cause complications in recipients. For this reason, we don’t need a full match when we use cord blood.”
The number of stem cells in one unit of umbilical cord blood, however, is not enough to repopulate the bone marrow of an adult. So after more mild chemo and radiation to kill any straggling cancer cells, Chakravarty underwent a double cord transplant, in which blood from two umbilical cords is combined to provide a sufficient number of stem cells.
The procedure is essentially painless, Shpall says, and is similar to a blood transfusion. The donated stem cells are thawed and infused into the recipient’s vein through an IV. The stem cells then naturally migrate into the bone marrow. The restored bone marrow begins producing normal blood cells within several days to several weeks.
“In some cases, the transplant can have an added benefit,” Shpall says. “The new blood cells also attack and destroy any cancer cells that survived the chemo and radiation.”
Age isn’t everything
While stem cell transplants may be lifesaving, not everyone is a candidate.
When approving a transplant, doctors take into account the patient’s overall physical condition, diagnosis, stage of disease and previous treatments. Tests are conducted to make sure the patient is healthy enough to undergo the procedure.
Stem cell transplants traditionally have been offered to children and young adults, though, ironically, blood cancers most often affect older patients.
“In the past, older adults had more complications with stem cell transplants, mainly due to the high levels of chemo and radiation required before the procedure,” Shpall explains. “Today, we’ve reduced the toxicity of these pre-treatment regimens.”
Doctors now are more likely to evaluate older patients on an individual basis, Shpall says, and to consider physiological age instead of chronological age when deciding who’s eligible for a transplant.
Taking it slowly
One month after receiving her donated stem cells, Chakravarty was released from the hospital.
Tests continue to show no signs of AML, but she knows her immune system may take months or even years to fully recover, so she’s “taking it slowly.”
This semester she's teaching a full course load, but online and hybrid classes only, where some traditional face-to-face “seat time” is replaced with online learning.
Chakravarty regularly returns to MD Anderson where doctors monitor her progress. She credits her family and friends, co-workers, MD Anderson caregivers, and devout Hindu faith with seeing her through.
“I view my cancer journey as a test,” she says. “This experience has deepened my belief in the goodness of humanity. Everyone supported me when I needed it most.”
How a stem cell transplant work
Certain cancers that affect the blood or immune system can devastate a person’s bone marrow, which is further destroyed by chemotherapy and radiation during treatment. When this happens, a stem cell transplant becomes necessary to replace the bone marrow. Here’s a basic breakdown of the process:
Stem cell transplant options
Different types of stem cell transplants are used to treat cancer. Their names are based on who donates the stem cells.
Autologous ("Auto")
This type of transplant uses a patient’s own stem cells, which are removed from their blood and frozen. When the patient is ready for transplant, the cells are thawed and given back to them.
Upside: Because the cells are a patient’s own, there’s less risk of rejection or graft-versushost disease, which occurs when a donor’s cells think the recipient’s cells are foreign and attacks them.
Downside: Some original cancer cells may remain in a patient’s donated supply.
Tandem (Double Autologous)
This is an autologous transplant performed twice instead of once, with a three- to six-month break in between. All the stem cells needed are collected beforehand, and half of them are used for each transplant.
Upside: Studies have shown improved survival rates and quality of life with tandem transplants compared to a single transplant.
Downside: Because two transplants are performed, the risk of complications is higher than for a single transplant.
Allogeneic (“Allo”)
Instead of using a patient’s own stem cells, those used in allogeneic transplants come from a donor. Stem cells from relatives or unrelated donors whose tissue type closely matches the patient’s may be used. Donors can be located through bone marrow registries such as the National Marrow Donor Program, and through umbilical cord blood banks.
Upside: The transplanted stem cells are cancerfree. Donor stem cells create a new immune system and may launch an attack against cancer cells remaining in the patient’s body.
Downside: A recipient’s body may see the donor stem cells as foreign and reject them, or the immune cells from the donor may attack not only cancer cells, but also healthy cells in the recipient’s body.
Mini (Reduced Intensity)
Patients receive less chemo and radiation before a mini-transplant than a standard transplant. The goal is to kill some, but not all, of the cancer cells and suppress the immune system just enough to allow donor stem cells to settle into the recipient’s bone marrow, take over, and launch an attack against the remaining cancer cells.
Upside: In the past, older patients and those who are too sick or frail to withstand the intense levels of chemo and radiation used in standard transplants were denied transplants. With the advent of mini-transplants, age and medical frailty are no longer “deal-breakers.”
Downside: Mini-transplants have only been performed since the late 1990s, so long-term patient outcomes are not yet clear. Though the procedure lowers the risk of some complications, cancer may be more likely to return.
Syngeneic (Identical Twin)
This type of allogeneic transplant can only be used if a patient has an identical sibling with the exact same tissue type.
Upside: Graft-versus-host disease will not be a problem.
Downside: Because a recipient’s new immune system is so much like their original, it can’t effectively fight the remaining cancer cells. Every effort must be made to destroy all the cancer cells before the transplant is done in order to help keep the cancer from coming back.
Half-matched
A half-matched donor is someone whose tissues are only half identical to the patient’s, but they’re close enough to allow him or her to move ahead with a transplant. Parents are always a half-match for their children, and vice versa. Siblings have a 50% chance of being a half-match for each other.
Upside: This procedure greatly expands the potential donor pool, making virtually all patients eligible for transplants.
Downside: Because the match between donor and recipient is only half, the donor’s immune system may generate a stronger attack against the recipient’s tissues.










